Introduction
It is estimated that 55% of the population aged 80 years and over have a disabling hearing loss [Roth et al., 2011], and 7–0.6% of the population aged 80 years and over have a bilateral severe to profound hearing loss, respectively, which does not benefit from conventional hearing aid amplification [Goman and Lin, 2016]. Furthermore, it is expected that the population aged over 85 years of age will increase markedly from 1.6 million in mid-2016 to 3.2 million by mid-2041 in the UK [Office for National Statistics, 2016].
Hearing loss inevitably leads to reduced speech discrimination and has been associated with multiple negative outcomes including social isolation [Weinstein, and Ventry, 1982; Mick et al., 2014; Sung et al., 2016], depression [Li et al., 2014], and reduced quality of life [Ciorba et al., 2012]. An association between peripheral hearing impairment and temporal lobe, as well as whole brain atrophy, has also been shown with neuroimaging studies [Lin et al., 2014]. Moreover, individuals with a mild to severe hearing loss have been shown to have a 2- to 5-fold increased risk, respectively, of developing dementia compared to those with normal hearing [Lin et al., 2011].
Cochlear implantation (CI) can successfully restore speech recognition for individuals with severe to profound sensorineural hearing loss for whom hearing rehabilitation using acoustic hearing aids has proved inadequate. Despite initial concerns, studies suggest that CI in the elderly is safe [Carlson et al., 2010; Migirov et al., 2010], has auditory rehabilitative outcomes comparable to those found for younger adults [Chatelin et al., 2004; Alice et al., 2013; Rohloff et al., 2017], and improves quality of life [Djalilian et al., 2002; Vermeire et al., 2005; Orabi et al., 2006; Cloutier et al., 2014]. Whether or not CI in the elderly can reduce cortical atrophy and the prevalence of dementia remains an area of ongoing interest [Völter et al., 2018].
Guidelines for CI are expanding, for instance in the UK, the National Institute for Health and Care Excellence (NICE) guidelines published in 2019 have expanded the candidacy for CI. This will further increase the number of CI procedures performed in the elderly population, far outpacing the growth in other hearing related surgeries. The duration of life expectancy over which an elderly patient may benefit from their CI will continue to be an area of interest, with particular relevance to cost-effectiveness. Consequently, this study investigates survival duration of CI recipients aged 80 years and over and discusses the cost utility of CI in this age group. In addition, the previous studies of surgical and audiological outcomes of CI in the elderly have either included smaller patient cohorts or recipients of less advanced ages. Therefore, secondary outcomes on post-operative complications are also presented as well as an analysis of speech discrimination outcomes.
Methods
Study Design
A retrospective study was undertaken of all CI recipients, aged 80 years and over at the time of their surgery, implanted at our institution (Cambridge University Hospitals) during a period from January 1, 2001, to March 31, 2019. Data were collected from January 1, 2001, to March 31, 2020, to allow for a minimum follow-up period of 1 year. Data were extracted from the audiometry database and electronic patient management system.
Participants and Data
Prior to CI, all patients received an audiological, speech and language, medical, and radiological assessment. In order to determine candidacy for CI, the audiological workup included pure-tone audiometry and a speech intelligibility evaluation. During the time window of this study, the criteria for CI were a bilateral sensorineural hearing loss of 90 dB or worse at 2 kHz and 4 kHz, with limited benefit from appropriately fitted hearing aids and a speech discrimination of 50% or worse assessed via the Bamford-Kowal-Bench (BKB) sentence test in quiet at 70 dBA in the best aided condition. Post CI, patients were enrolled onto a rehabilitation and programming schedule, which comprised initial implant activation at the fourth week post-surgery, followed by an intensive 1 month of programming sessions with hearing therapy and assessment of functional outcomes at 2 months and 12 months.
In total, data were collected for 122 patients and 126 surgeries. Four patients were explanted and re-implanted on the same side during the time course of this study, of which 3 were initially implanted during the study period. One patient underwent sequential implantation. The medical data collected included patient demographics, age at CI surgery, side, device type, American Society of Anesthesiologists (ASA) score, anaesthetic type, anaesthetic and surgical complications, and survival duration. The audiological data collected included pure-tone average (pre- and at the second month post-implantation) and speech perception scores for the BKB sentence test in quiet and in noise (pre- and at the second and twelfth months post-implantation). The BKB sentence test comprised of recorded open set sentences, presented in a sound field at 70 dBA with male and female voice in both quiet and noise (using pink noise at 10 dBA level). Data were analysed using MATLAB (MathWorks, Natick, MA). Mean and standard deviation were used unless otherwise specified. Statistical analyses were performed using the R software package (http://www.r-project.org). Values of p < 0.05 were considered to be statistically significant.
Statistical Analysis
Survival times were calculated from the date of surgery to the occurrence of either death or the end of the follow-up period (March 31, 2020). For patients, who were explanted and re-implanted during the course of this study, survival times were calculated from the date of the most recent surgery. Survival curves were generated separately for male and female patients using the Kaplan-Meier method [Cardillo, 2020]. The hazard rate is defined as the average rate of death per year.
The effects of age, gender, pre-implantation BKB scores, and ASA grade on speech discrimination outcomes were examined by fitting separate linear models to the post-implantation BKB scores measured at 2 and 12 months. For the analysis, data from 39 out of 126 cases were excluded due to the following criteria:
Re-implantation surgery (4)
Sequential implantation (1)
Ineraid user (1)
Diagnosed with dementia soon after the cochlear implant (3)
English as a second language (2)
British Sign Language users (2)
Non-traditional recipients (3)
Health deteriorated prior to testing (5)
Without speech score data at 2 and 12 months (18)
In total, data from 87 cases were included in the analysis, of which 76 patients had a post-implantation BKB score at 2 months (i.e., 50/126 cases excluded), and 60 patients had a post-implantation BKB score at 12 months (i.e., 66/126 cases excluded). A preliminary analysis of the post-implantation BKB scores showed that the data were negatively skewed. Therefore, prior to statistical analysis, a reflect and square transformation was applied. The model fit was checked using the Shapiro-Wilk test (2 months: W = 0.98, p value = 0.24; 12 months: W = 0.98, p value = 0.29) and by generating Q-Q plots, which both showed that the residuals were normally distributed and therefore confirmed the model fit.
Results
Demographics
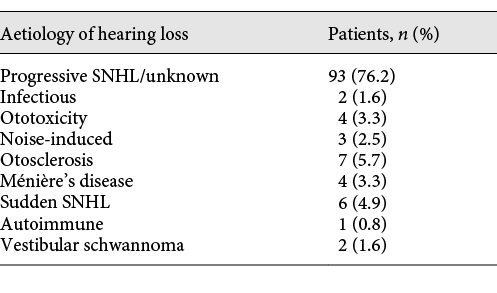
A total of 122 patients (126 surgeries) aged 80 years and older were included in this study. The aetiology of hearing loss is listed in Table 1. The mean age at CI surgery was 84 years (SD = 3.6 years, shown in Fig. 1a, b). Over the time course of this study, the number of CI performed in adults aged 80 years and older showed an increasing trend (shown in Fig. 1c), which reflects a change in the NICE guidelines that increased candidacy in 2009.
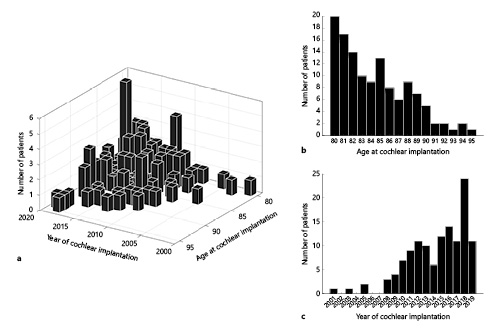
Fig. 1
Age and year of CI. a Distribution of age at CI according to year of CI. b, c Individual distributions of age at CI (b) and year of CI (c). Data shown from January 1, 2001, to March 31, 2019 (n = 126). CI, cochlear implantation.
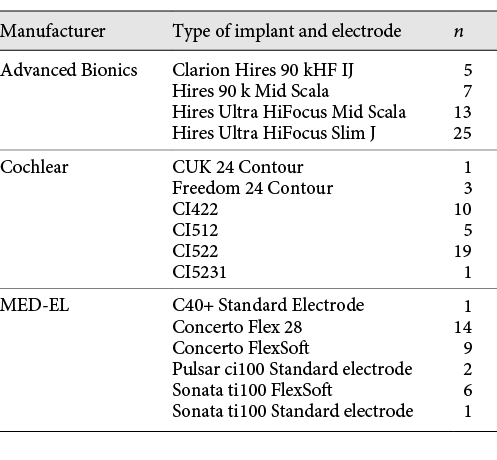
Patients were implanted with devices from Advanced Bionics (n = 50; 41%), Cochlear (n = 39; 32%), and MED-EL (n = 33; 27%) (shown in Table 2). Due to multiple co-morbidities, 1 patient was implanted under local anaesthesia. The remainder of patients were implanted under general anaesthesia. Four patients were reported to have partial insertion of the electrode array. The right ear was implanted in 66 (56%) cases and the left ear in 56 (44%) cases. The mean pure-tone average pre-implantation was 108 dB HL (SD = 14.6 dB HL), and the mean BKB score pre-implantation was 14% (SD = 17%). All patients reported to use their CI during the first year of the follow-up. The median ASA grade was 2.2 (SD = 0.81).
Survival Duration
The cohort of 122 patients was divided into 2 subgroups based on their status at March 31, 2020: 38 deceased patients (31%) and 84 surviving patients (69%). The mean length of survival with the cochlear implant for the deceased group was 49 months (SD = 37 months). Eight patients in the deceased group used their implants for more than 6 years (shown in Fig. 2a). The mean length of survival in the surviving group was 47 months (SD = 32 months). This includes patients who had been implanted as recently as the preceding year (shown in Fig. 2b). The survival probability of all CI recipients at 12 months post-operatively was 0.95 for females and 0.93 for males, at 36 months post-operatively was 0.89 for females and 0.81 for males, and at 60 months post-operatively was 0.74 for females and 0.54 for males (shown in Fig. 2c). The average hazard rate for females was 0.04 and for males was 0.02.
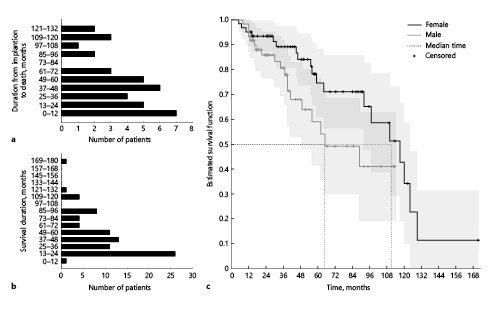
Fig. 2
Survival duration post CI. a Distribution of time from CI to death (n = 38). b Distribution of time from CI to March 31, 2020, in surviving patients (n = 84). c Survival curve generated using the Kaplan-Meier method for female (black line) and male (grey line) patients (n = 122). Dotted lines show the median survival time. Dashes indicate censored data. CI, cochlear implantation.
Audiological Outcomes
Seventy-six patients had a BKB score in quiet at 2 months post-implantation, and 60 patients had a BKB score in quiet at 12 months post-implantation (see methods for exclusion criteria). The mean BKB score at 2 and 12 months post-implantation was 66% (SD = 27%, N = 76) and 72% (SD = 21%, N = 60), respectively. The mean gain in BKB score from pre-implantation was 51% (SD = 30%) at 2 months post-implantation and 58% (SD = 26%) at 12 months post-implantation. An analysis of the effect of implant type (Advanced Bionics, Cochlear, and MED-EL) on speech discrimination scores (shown in Fig. 3) did not reveal any significant difference at either 2 months (Kruskal-Wallis test: H74 = 1.42, p = 0.49) or 12 months (H58 = 3.81, p = 0.15).
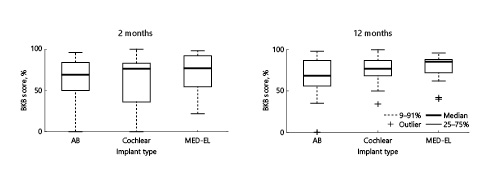
Fig. 3
The effect of implant type on speech discrimination outcomes. Boxplots showing Bamford-Kowal-Bench (BKB) scores in quiet at 2 months (left column) and 12 months (right column) post-operatively for Advanced Bionics (AB), Cochlear, and MED-EL devices.
In order to investigate the effects of age at implantation, gender, and pre-implantation BKB scores on post-implantation BKB scores measured at 2 and 12 months (shown in Fig. 4), separate linear models were fitted to the data. At 12 months, the model showed that BKB scores decreased significantly with increasing age at implantation (t51 = 1.98, p = 0.05) and ASA grade (1 vs. 2: t51 = 2.00, p = 0.05; 1 vs. 3: t51 = 2,13, p = 0.04; 1 vs. 4: t51 = 3.00, p < 0.01), whereas at 2 months although a similar trend was found, for both age (t67 = 1.58, p = 0.12) and ASA grade (1 vs. 2: t67 = 0.59, p = 0.56; 1 vs. 3: t67 = 0.12, p = 0.90; 1 vs. 4: t67 = −0.64, p = 0.52), this was not statistically significant. At both 2 and 12 months, the models showed that neither gender (2 months: t67 = −1.32, p = 0.19; 12 months: t51 = −1.87, p = 0.07) nor pre-implantation BKB scores (2 months: t72 = −0.92, p = 0.36; 12 months: t51 = −0.52, p = 0.61) significantly affected post-implantation BKB scores.
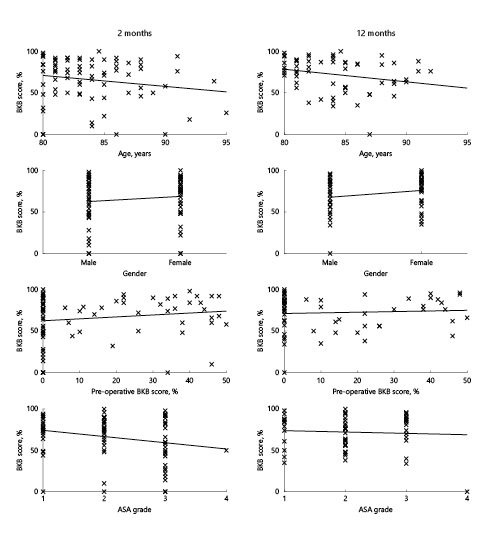
Fig. 4
Speech discrimination outcomes. Scatter plots showing Bamford-Kowal-Bench (BKB) scores in quiet at 2 months (left column) and 12 months (right column) post-operatively, according to the age at implantation (years), gender, BKB score pre-operatively (%), and American Society of Anesthesiologists (ASA) grade. Scatter plots are shown with a least squares fit line.
Three patients were diagnosed with dementia post-CI, which precluded quantitative measurement of speech discrimination performance. However, for 2 of these patients, a qualitative measure, in the form of a Categories of Auditory Performance (CAP) score, was available. One patient was able to recognise environmental sounds (CAP-3) and the other understand conversation without lip-reading with a familiar talker (CAP-6). Of the 5 patients who were unable to complete testing due to health reasons, 1 patient had a stroke at 2 months post-CI, 1 patient was diagnosed with Parkinson’s disease, and the remaining 3 patients experienced a general decline in their health, with death occurring between 7 and 13 months post-CI.
Complications
Patient records were interrogated for reported complications. Two cases out of 126 had inaccessible records and consequently, the peri-operative notes were analysed for 124 cases. The mean ASA grade was 2.1 (SD = 0.81). Many patients had chronic diseases including hypertension (n = 45), atrial fibrillation (n = 24) coronary artery disease (n = 19), diabetes mellitus (n = 10), chronic obstructive pulmonary disease (n = 6), and chronic kidney disease (n = 3). Peri-operative and long-term complications were divided into anaesthetic, surgical, and device related outcomes.
Overall, 19 patients (15.3%) experienced complications. No major cardiac, pulmonary, or neurologic anaesthetic complications were reported. In the immediate post-operative period, 2 patients (1.6%) developed delirium lasting for less than 24 h, and 1 patient developed diarrhoea (0.8%). No CI recipient required intensive care unit monitoring; however, 1 patient required prolonged hospital admission for urinary retention, hyponatraemia, and hypothyroidism. Two patients (1.6%) described taste alteration, 1 reported new or worsening tinnitus, 8 patients (6.4%) reported new or worsening diziness lasting for less than 1 month, and 2 patients (1.6%) reported persistent dizziness lasting for more than 1 month. Two patients developed wound infections that settled with oral antibiotics (1.6%). No cases of facial nerve paresis were reported. The average length of stay was 1.1 nights (SD = 1.6), and 16 (12.9%) patients were discharged the same day. The ASA score (2.1 vs. 2.1, t117 = −0.09, p = 0.93) and patient age (83.9 vs. 84.6 years, t117 = −0.67, p = 0.50) did not differ significantly between the day surgery and overnight stay groups.
Over the time course of this study, 4 patients (3.3%) were explanted and re-implanted on the same side. The first patient was re-implanted due to infection. The second patient developed a scalp pneumocele after vigorous nose blowing and experienced a reduction cochlear implant function, secondary to electrode displacement. The third patient, after coughing, also experienced a reduction cochlear implant function, secondary to electrode displacement. Finally, the fourth patient was re-implanted due to mis-placement of the initial electrode array.
Discussion
The expansion of CI candidacy together with the exponential rise in patients living over 80 years have increased the number of elderly CI recipients and consequently has brought to the forefront concerns regarding the efficacy, safety, and cost utility of CI in this age group. This study presents surgical and audiological outcomes for the largest population of elderly CI recipients, to date, and is also the first to investigate survival duration in this cohort. Our results show that CI in patients aged 80 years and over is associated with significantly improved auditory function and although surgery can be associated with complications, particularly vestibular, life threatening complications are rare, and CI in this age group is typically a safe procedure.
Over the last 15 years, several units have published studies examining speech discrimination scores in elderly patients in comparison to younger patients [Chatelin et al., 2004; Leung et al., 2005; Orabi et al., 2006; Kayser et al., 2008; Carlson et al., 2010; Dillon et al., 2013; Mosnier et al., 2014; Jolink et al., 2016; Wong et al., 2016]. The majority have shown that there is no difference in functional outcomes between elderly and young patient cohorts and that duration of deafness is a more significant negative factor than age [Leung et al., 2005; Mosnier et al., 2014]. This has likely contributed to an increasing global trend to offer CI to elderly patients, and such a trend is seen in our unit. Our results show that CI in patients aged 80 years or older is associated with significantly improved auditory function as evidenced by the gain in BKB scores pre- and post-implantation, and likewise, we found no significant effect of age at implantation or ASA grade on BKB scores at 2 months post-implantation.
Speech discrimination improved between 2 and 12 months post-operatively, indicating an auditory rehabilitative benefit and is consistent with findings from other studies looking at CI outcomes amongst the elderly population [Leung et al., 2005; Budenz et al., 2011; Clark et al., 2012; Lazard et al., 2012; Rohloff et al., 2017]. Evidence suggests that speech perception scores continue to improve to up to 5 years post-implantation [Dillon et al., 2013] and can remain stable over an average of 8.4 years. The resultant outlook is an overall improved quality of life [Huarte et al., 2014], which has been shown to be long-lasting. In addition, CI has been show to improve cognitive performance, for example, Mosnier et al. [2014] found that 81% of patients with poor performance showed improved performance scores at 1-year post-implantation with a linear relationship between speech perception and cognitive function over time. Furthermore, positive outcomes have also been noted in mental health status, for example, Choi et al. [2016] noted a significant reduction in depression 1-year post-implantation. The social independence afforded by CI is also an important consideration against the cost of residential care for the growing elderly population.
However, our results also show that at 12 months post-implantation BKB scores decreased significantly with increasing age at implantation and ASA grade. This result may reflect age-related changes such as a reduction in the number of spiral ganglion cells, central presbyacusis, and a reduction in the capacity for central nervous system plasticity and highlights a number of areas to consider surrounding CI in the elderly, which are potentially more pertinent than for younger candidates. Issues surrounding auditory cortex plasticity and rehabilitation in the ageing brain have been contentious. Schierholz et al. [2015] used electroencephalogram to examine the audiovisual interactions in the auditory cortex of elderly CI recipients and found that elderly patients had stronger visual modulation of their auditory cortex in comparison to younger CI users and individuals with normal hearing. The authors postulate that the process of ageing, sensory deprivation, and possibly CI experience had resulted in over-reliance on the other senses and potential coupling of the auditory and visual modalities. Consequently, a multisensory rehabilitation strategy, relying more on audiovisual input may be an area to explore for auditory rehabilitation in elderly patients. Additionally, comparison of speech recognition score outcomes between 2 groups of patients with mean auditory deprivation time of 2.52 years and 19.67 years showed no difference between the 2 groups, suggesting that the time lapse in post-lingual hearing loss should not be a factor in the decision to implant elderly patients [Medina et al., 2017].
Previously, concerns have been raised regarding the ability to rehabilitate and train an elderly CI user, particularly in the context of potential multiple comorbidities and functional limitations [Schumann et al., 2014]. The authors stress that regular auditory training is essential in the rehabilitation process and has a major role to play based on the belief that the plasticity of the central auditory system allows for improvement in speech perception in everyday listening. It is also recognised that psychological health is also an important factor in the success of implantation in the elderly. Knopke et al. [2016] found that stress, anxiety, and depression impacted negatively on quality of life outcomes in elderly CI patients.
This is the first study to investigate the length of time elderly CI recipients go on to live with their implant. The mean length of survival with a CI for the deceased group was 49 months. When the entire study population was included, the probability of a female and male CI recipient aged 80 years or older being alive at 3 years was 0.89 and 0.81, respectively and at 5 years was 0.74 and 0.54. Our longest surviving CI recipient was still using their implant after more than 13 years having been implanted at the age of 85 years. Our complication rate was 15.3%, which is consistent with other reported series of older and younger patients [Chen et al., 2013; Wong et al., 2016; Yang, and Cosetti, 2016]. The commonest complication reported was vestibular disturbance, which affected 8% of patients and requires particular attention in this age group due to the increased prevalence of pre-existing vestibular, proprioceptive, and visual deficits and the associated risk of falls. However, after 1 month, 8 out of the 10 patients with vestibular disturbance reported symptom resolution. Previous studies have shown the prevalence of post-implantation dizziness to be as high as 39% in adult patients with predictive factors including presence of pre-operative dizziness, age at implantation, and age at onset of hearing loss [Fina et al., 2003]. The general consensus is that the short and long term benefits of CI are likely to outweigh the potential risks of surgery, which are rarely life-threatening, in motivated elderly individuals [Schumann et al., 2014].
The cost-utility of CI in the elderly remains an area of interest. A study undertaken for the National Institution of Health Research Health Technology Assessment programme in the UK found that the incremental cost-effectiveness ratio for unilateral implantation compared with no implantation is GBP 14,163 per quality-adjusted life year (QALY) and that unilateral implantation confers 0.075 QALYs per year over an average 32-year period [Bond et al., 2009]. Figures from the Office for National Statistics [Office for National Statistics, 2020] show that a 50-year-old male in the UK can expect to live on average another 31 years. With an estimated lifetime cost of CI totalling GBP 34,000, this would mean a cost of GBP 14,624 per QALY using the calculations in the National Institution of Health Research study. For a male aged 80, average life expectancy is 8 years. Using a lower overall tariff of GBP 31,000 to account for fewer maintenance and repair costs, the cost per QALY for this age group would nevertheless be GBP 51,667 per QALY. For a 90-year-old, the equivalent calculation comes to GBP 93,000 per QALY.
The NICE makes decisions regarding the cost-effectiveness of treatments offered by the National Health Service in the UK In general interventions with an incremental cost-effectiveness ratio of less than GBP 20,000, per QALY gained are considered to be cost-effective. The calculations for older patients would therefore exceed this threshold. However, it is argued that cost-effectiveness evaluation using QALYs is discriminatory to older population who naturally have a shorter life expectancy [Tsuchiya, 2000] and in line with most healthcare systems in developed countries, the National Health Service does not discriminate by age. In addition, the above calculations do not consider other factors that could affect cost-utility, such as prevention of dementia and more independent community-based function. It should also be kept in mind that communication ability might make the difference in older individuals between living independently and needing more care from other societal resources, also significantly mitigating the costs of implantation.
Our study is of retrospective nature, examining a window of time during which the tendency to offer implantation to elderly patients has changed. As this patient group evolves, it will be valuable to examine the cohort in a prospective fashion whilst also increasing the sample size from that which is currently available. Further trends may emerge as cochlear implant candidacy widens and patients and their referrers become more aware that this surgery is available to them. However, our results suggest that CI in patients aged 80 years and over is well-tolerated with a low complication rate and is associated with significantly improved auditory function, and consequently, routine implantation in this age group should not be overlooked.
Statement of Ethics
This study protocol was reviewed and approved by Cambridge University Hospitals Research and Audit Department, approval No. 3768.
Conflict of Interest Statement
The authors have no conflicts of interest to declare.
Funding Sources
The authors have no sources of funding to declare.
Author Contributions
A.H.K. contributed to data analysis, statistical analysis, manuscript drafting, and editing. D.B. contributed to manuscript revision and study design. J.M. contributed to manuscript drafting and editing. T.P. contributed to data collection. A.V. contributed to manuscript drafting and editing. M.B. contributed to manuscript revision. J.T contributed to manuscript revision. P.A. contributed to manuscript revision. N.D. contributed to manuscript revision and study design.
References
- 1. Alice B, Silvia M, Laura G, Patrizia T, Roberto B. Cochlear implantation in the elderly: surgical and hearing outcomes. BMC Surg. 2013;13(Suppl 2):S1.http://dx.doi.org/10.1186/1471-2482-13-S2-S1.
- 2.
- 3. Bond M, Mealing S, Anderson R, Elston J, Weiner G, Taylor RS, et al. The effectiveness and cost-effectiveness of cochlear implants for severe to profound deafness in children and adults: a systematic review and economic model. Health Technol Assess. 2009;13(44):1–330.
- 4. Budenz CL, Cosetti MK, Coelho DH, Birenbaum B, Babb J, Waltzman SB, et al. The effects of cochlear implantation on speech perception in older adults. J Am Geriatr Soc. 2011;59(3):446–53.http://dx.doi.org/10.1111/j.1532-5415.2010.03310.x.
- 5.
- 6. Carlson ML, Breen JT, Gifford RH, Driscoll CL, Neff BA, Beatty CW, et al. Cochlear implantation in the octogenarian and nonagenarian. Otol Neurotol. 2010;31(8):1343–9.http://dx.doi.org/10.1097/MAO.0b013e3181edb69d.
- 7. Chatelin V, Kim EJ, Driscoll C, Larky J, Polite C, Price L, et al. Cochlear implant outcomes in the elderly. Otol Neurotol. 2004;25(3):298–301.http://dx.doi.org/10.1097/00129492-200405000-00017.
- 8. Chen DS, Clarrett DM, Li L, Bowditch SP, Niparko JK, Lin FR. Cochlear implantation in older adults: long-term analysis of complications and device survival in a consecutive series. Otol Neurotol. 2013;34(7):1272–7.http://dx.doi.org/10.1097/MAO.0b013e3182936bb2.
- 9. Choi JS, Betz J, Li L, Blake CR, Sung YK, Contrera KJ, et al. Association of using hearing aids or cochlear implants with changes in depressive symptoms in older adults. JAMA Otolaryngol Head Neck Surg. 2016;142(7):652.http://dx.doi.org/10.1001/jamaoto.2016.0700.
- 10. Ciorba A, Bianchini C, Pelucchi S, Pastore A. The impact of hearing loss on the quality of life of elderly adults. Clin Interv Aging. 2012;7:159.http://dx.doi.org/10.2147/CIA.S26059.
- 11. Clark JH, Yeagle J, Arbaje AI, Lin FR, Niparko JK, Francis HW. Cochlear implant rehabilitation in older adults: literature review and proposal of a conceptual framework. J Am Geriatr Soc. 2012;60(10):1936–45.http://dx.doi.org/10.1111/j.1532-5415.2012.04150.x.
- 12. Cloutier F, Bussières R, Ferron P, Côté M. OCTO “Outcomes of cochlear implant for the octogenarians: audiologic and quality-of-life”. Otol Neurotol. 2014;35:22–8.
- 13. Dillon MT, Buss E, Adunka MC, King ER, Pillsbury HC, Adunka OF, et al. Long-term speech perception in elderly cochlear implant users. JAMA Otolaryngol Head Neck Surg. 2013;139(3):279.http://dx.doi.org/10.1001/jamaoto.2013.1814.
- 14. Djalilian HR, King TA, Smith SL, Levine SC. Cochlear implantation in the elderly: results and quality-of-life assessment. Ann Otol Rhinol Laryngol. 2002;111(10):890–5.http://dx.doi.org/10.1177/000348940211101005.
- 15. Fina M, Skinner M, Goebel JA, Piccirillo JF, Neely JG, Black O. Vestibular dysfunction after cochlear implantation. Otol Neurotol. 2003;24(2):234–42; discussion 242.http://dx.doi.org/10.1097/00129492-200303000-00018.
- 16. Goman AM, Lin FR. Prevalence of hearing loss by severity in the United States. Am J Public Health. 2016;106(10):1820–2.http://dx.doi.org/10.2105/AJPH.2016.303299.
- 17. Huarte A, Lezaun R, Manrique M. Quality of life outcomes for cochlear implantation in the elderly. Audiol Neurotol. 2014;19(1):36–9.http://dx.doi.org/10.1159/000371608.
- 18. Jolink C, Helleman HW, van Spronsen E, Ebbens FA, Ravesloot MJ, Dreschler WA. The long-term results of speech perception in elderly cochlear implant users. Cochlear Implants Int. 2016;17(3):146–50.http://dx.doi.org/10.1080/14670100.2016.1162383.
- 19. Kayser C, Petkov CI, Logothetis NK. Visual modulation of neurons in auditory cortex. Cereb Cortex. 2008;18(7):1560–74.http://dx.doi.org/10.1093/cercor/bhm187.
- 20. Knopke S, Gräbel S, Förster-Ruhrmann U, Mazurek B, Szczepek AJ, Olze H. Impact of cochlear implantation on quality of life and mental comorbidity in patients aged 80 years. Laryngoscope. 2016;126(12):2811–6.http://dx.doi.org/10.1002/lary.25993.
- 21. Lazard DS, Vincent C, Venail F, Van de Heyning P, Truy E, Sterkers O, et al. Pre-, per- and postoperative factors affecting performance of postlinguistically deaf adults using cochlear implants: a new conceptual model over time. PLoS One. 2012;7(11):e48739.http://dx.doi.org/10.1371/journal.pone.0048739.
- 22. Leung J, Wang NY, Yeagle JD, Chinnici J, Bowditch S, Francis HW, et al. Predictive models for cochlear implantation in elderly candidates. Arch Otolaryngol Head Neck Surg. 2005;131(12):1049.http://dx.doi.org/10.1001/archotol.131.12.1049.
- 23. Li CM, Zhang X, Hoffman HJ, Cotch MF, Themann CL, Wilson MR. Hearing impairment associated with depression in US adults, National Health and Nutrition Examination Survey 2005–2010. JAMA Otolaryngol Head Neck Surg. 2014;140(4):293.http://dx.doi.org/10.1001/jamaoto.2014.42.
- 24. Lin FR, Ferrucci L, An Y, Goh JO, Doshi J, Metter EJ, et al. Association of hearing impairment with brain volume changes in older adults. Neuroimage. 2014;90:84–92. http://dx.doi.org/10.1016/j.neuroimage.2013.12.059.
- 25. Lin FR, Ferrucci L, Metter EJ, An Y, Zonderman AB, Resnick SM. Hearing loss and cognition in the Baltimore Longitudinal Study of Aging. Neuropsychology. 2011;25(6):763–70.http://dx.doi.org/10.1037/a0024238.
- 26. Medina MDM, Polo R, Gutierrez A, Muriel A, Vaca M, Perez C, et al. Cochlear implantation in postlingual adult patients with long-term auditory deprivation. Otol Neurotol. 2017;38(8):e248–52.http://dx.doi.org/10.1097/MAO.0000000000001257.
- 27. Mick P, Kawachi I, Lin FR. The association between hearing loss and social isolation in older adults. Otolaryngol Head Neck Surg. 2014;150(3):378–84.http://dx.doi.org/10.1177/0194599813518021.
- 28. Migirov L, Taitelbaum-Swead R, Drendel M, Hildesheimer M, Kronenberg J. Cochlear implantation in elderly patients: surgical and audiological outcome. Gerontology. 2010;56(2):123–8.http://dx.doi.org/10.1159/000235864.
- 29. Mosnier I, Bebear J-P, Marx M, Fraysse B, Truy E, Lina-Granade G, et al. Predictive factors of cochlear implant outcomes in the elderly. Audiol Neurotol. 2014;19(1):15–20.http://dx.doi.org/10.1159/000371599.
- 30.
- 31.
- 32.
- 33. Orabi AA, Mawman D, Al-Zoubi F, Saeed SR, Ramsden RT. Cochlear implant outcomes and quality of life in the elderly: Manchester experience over 13 years. Clin Otolaryngol. 2006;31(2):116–22.http://dx.doi.org/10.1111/j.1749-4486.2006.01156.x.
- 34. Rohloff K, Koopmann M, Wei D, Rudack C, Savvas E. Cochlear implantation in the elderly: does age matter?Otol Neurotol. 2017;38(1):54–9.http://dx.doi.org/10.1097/MAO.0000000000001262.
- 35. Roth TN, Hanebuth D, Probst R. Prevalence of age-related hearing loss in Europe: a review. Eur Arch Otorhinolaryngol. 2011;268(8):1101–7.http://dx.doi.org/10.1007/s00405-011-1597-8.
- 36. Schierholz I, Finke M, Schulte S, Hauthal N, Kantzke C, Rach S, et al. Enhanced audio-visual interactions in the auditory cortex of elderly cochlear-implant users. Hear Res. 2015;328:133–47.http://dx.doi.org/10.1016/j.heares.2015.08.009.
- 37. Schumann A, Hast A, Hoppe U. Speech performance and training effects in the cochlear implant elderly. Audiol Neurotol. 2014;19(1):45–8.http://dx.doi.org/10.1159/000371611.
- 38. Sung YK, Li L, Blake C, Betz J, Lin FR. Association of hearing loss and loneliness in older adults. J Aging Health. 2016;28(6):979–94.http://dx.doi.org/10.1177/0898264315614570.
- 39. Tsuchiya A. QALYs and ageism: philosophical theories and age weighting. Health Econ. 2000;9(1):57–68.http://dx.doi.org/10.1002/(sici)1099-1050(200001)9:1<57::aid-hec484>3.0.co;2-n.
- 40. Vermeire K, Brokx JP, Wuyts FL, Cochet E, Hofkens A, Van de Heyning PH. Quality-of-life benefit from cochlear implantation in the elderly. Otol Neurotol. 2005;26(2):188–95.http://dx.doi.org/10.1097/00129492-200503000-00010.
- 41. Völter C, Götze L, Dazert S, Falkenstein M, Thomas JP. Can cochlear implantation improve neurocognition in the aging population?Clin Interv Aging. 2018;13:701–12.
- 42. Weinstein BE, Ventry IM. Hearing impairment and social isolation in the elderly. J Speech Hear Res. 1982;25(4):593–9.http://dx.doi.org/10.1044/jshr.2504.593.
- 43. Wong DJY, Moran M, O’Leary SJ. Outcomes after cochlear implantation in the very elderly. Otol Neurotol. 2016;37(1):46–51.http://dx.doi.org/10.1097/mao.0000000000000920.
- 44. Yang Z, Cosetti M. Safety and outcomes of cochlear implantation in the elderly: a review of recent literature. J Otol. 2016;11(1):1–6.http://dx.doi.org/10.1016/j.joto.2016.03.004