Introduction
The pulmonary artery catheter, also known as the Swan-Ganz catheter, was introduced into clinical practice in 1970 []. It was, historically, the first device capable of categorizing the hemodynamic profile of critically ill patients and it has been widely used both as a diagnostic tool and to guide therapy [].
The use of pulmonary artery catheterization (PAC) was questioned after some observational studies suggested that it is associated with increased mortality [,]. Subsequently, randomized clinical trials (RCT) [,,,,] and meta-analysis [] failed to demonstrate any clinical benefit. Since then, its use has progressively declined [,]. It is now accepted that the routine use of PAC in critically ill patients is ineffective. Nevertheless, PAC still has an important role in patients with acute heart failure and current guidelines suggest it should be considered for such patients who are persistently hypotensive [,,]. Besides, in patients with cardiogenic shock (CS), PAC is still extensively used [] and even recommended [] to confirm the diagnosis and to guide changes in therapy. Despite this widespread practice, previous studies that investigated the effectiveness of PAC in critically ill patients did not specifically address its effect on patients with CS []. Thus, the influence of PAC on CS patient survival is not well established and its use remains controversial. We aimed to assess the impact of the PAC on short- and long-term clinical outcomes in a contemporary cohort of CS patients.
Methods
Study Design
We performed a prospective cohort investigation of all consecutive patients who were admitted to our intensive cardiac care unit (ICCU) from December 2005 to May 2009 presenting with a first admission of CS. The patient flow chart is shown in figure 1. Baseline clinical characteristics, complications and the use of PAC or other procedures during the admission period were collected on a daily basis until the resolution of the index episode. The study was conducted in accordance with the standards set by the Declaration of Helsinki and was approved by the Clinical Research Ethics Committee at our institution.
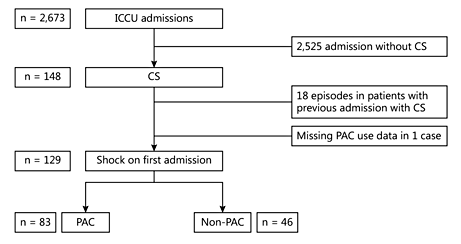
Fig. 1
Study flow diagram.
Definitions and Study Protocol
No formal management protocols were used in this study. Locating the venous access, which device to use and the subsequent clinical management were decided on at the discretion of the treating clinicians. The decision to remove or replace the device was at the discretion of the ICCU team.
CS criteria included systolic blood pressure <90 mm Hg (in the absence of hypovolemia and after adequate fluid challenge) for 30 min or the need for vasopressor therapy to maintain adequate perfusion pressure and signs of hypoperfusion (≥2 of the following: altered mental status/confusion, cold periphery and oliguria). Patients with CS were categorized into those who presented with CS upon admission and those who developed CS during their admission, having initially been admitted for other causes.
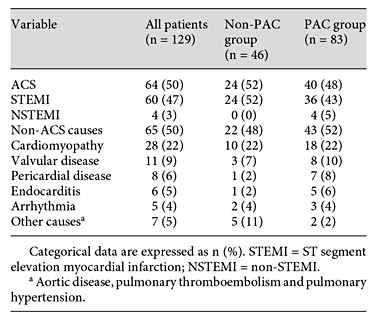
The causes of CS were classified as: due to acute coronary syndrome (ACS) or non-ACS. ACS included ST- and non-ST segment elevation myocardial infarction, and was defined according to the universal definition of myocardial infarction []. In all ACS cases, the presence and location of acute coronary occlusion was documented by coronary angiography. Non-ACS causes of CS included valvular disease, cardiomyopathy, pericardial disease, arrhythmia and endocarditis.
Outcome Measures
The primary end point was the assessment of 30-day all-cause mortality and long-term mortality adjusted for clinically relevant and statistically significant variables. A secondary end point, comparing short- and long-term mortality in patients with and without ACS upon admission was also recorded.
Statistical Analysis
The baseline characteristics of the 2 groups (PAC vs. non-PAC) were compared with the use of the χ2 test, Fisher exact or Student t test. All tests were 2-sided. Univariate and multivariate analyses for 30-day and long-term mortality were performed, using the Cox proportional-hazards regression model, to the estimate hazard ratio (HR) and 95% confidence interval (CI). The final models were adjusted for those variables categorized as clinically relevant with previous studies supporting their prognostic importance, such as age, gender and presence of CS upon admission []. The adjusted number needed to treat (NNT), i.e. the expected number of subjects who would need to undergo PAC to yield 1 additional good outcome, compared with the non-PAC group, was also calculated for both Cox models. Overall survival curves were obtained using the Kaplan-Meier method and compared using the log-rank test. Subgroup study of 30-day mortality on patients with and without ACS upon admission was assessed by means of the χ2 test and by adding an interaction (ACS*PAC) in both final Cox models. p < 0.05 was considered statistically significant. The analyses were performed using the statistical package SPSS for Windows (SPSS Statistics v21, IBM, Armonk, N.Y., USA).
STATA software v13.1 (Stata Corp., College Station, Tex., USA) was used to assess the performance of the models. The proportional-hazards assumption was tested using Schoenfeld residuals. Calibration (overall goodness of fit) for the Cox proportional-hazards model was tested by means of the Grønnesby and Borgan test. The prognostic accuracy of the model was quantified using the Harrell concordance (c)-statistic. Finally, the internal validity of the model was assessed by the bootstrap resampling technique, using the Harrell ‘rms' package in the R project for statistical computing. Results were reported according to the research reporting guidelines for observational studies (the STROBE statement) [].
Results
Study Population
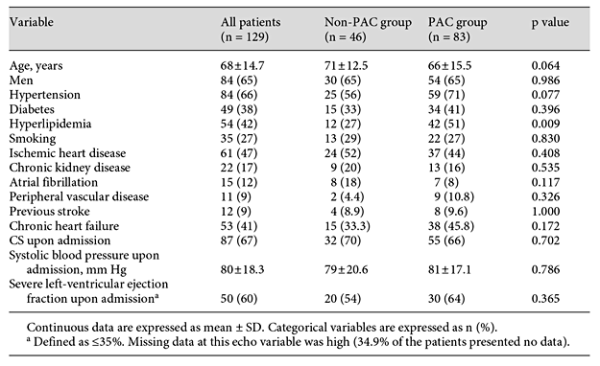
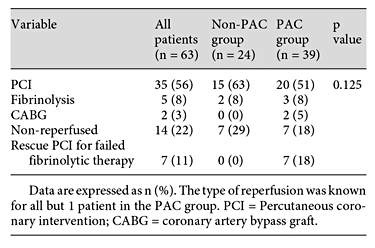
Baseline characteristics (i.e. upon admission) did not differ between patients with and without PAC, with the exception of hyperlipidemia (table 1). CS was due to non-ACS conditions in 50% of the patients with ACS accounting for the remaining 50% (table 2). The type of reperfusion in patients with ACS is reported in table 3.
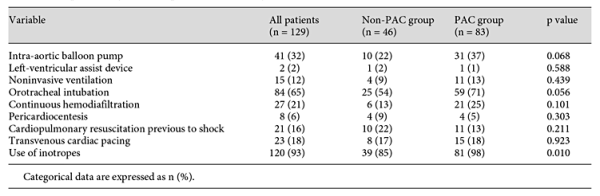
PAC was used in 64% of all patients, with a well-balanced distribution between those with ACS and those without ACS (PAC was used in 48% of ACS patients compared to 52% of non-ACS cases, p = 0.665). Inhospital management data and other procedures are illustrated in table 4. Noteworthy is that inotropes were used more frequently in the PAC group. Uses of intra-aortic balloon pump and orotracheal intubation were also higher in this group, approaching but not reaching significance.
Short-Term Mortality
Overall mortality at day 30 was 64% (n = 82). Patients with PAC had decreased 30-day mortality compared with non-PAC patients (55 vs. 78%; p = 0.010). The crude HR for mortality in the PAC group was 0.45 (95% CI 0.31 - 0.76, p = 0.001). Figure 2a shows the Kaplan-Meier survival curves.
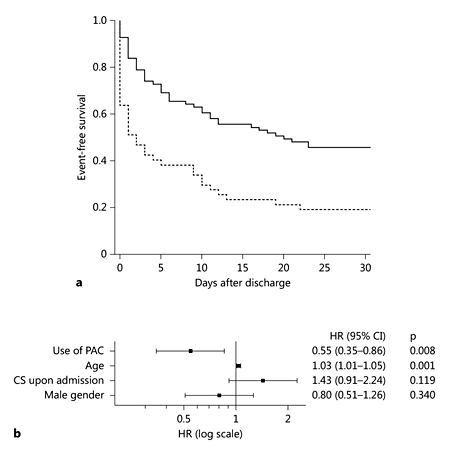
Fig. 2
Impact of PAC on 30-day mortality. a Kaplan-Meier survival estimates during 30 days. The solid line indicates the PAC group and the dashed line indicates the non-PAC group. b HR and 95% CI estimates of variables were included in the adjusted multivariate model to assess 30-day mortality.
PAC remained an independent 30-day predictor of survival after adjustment for age, gender and the presence of CS upon admission (compared to those who developed CS during hospitalization but not presenting the disorder at admission). The adjusted HR for death in the PAC group compared with the non-PAC group was 0.55 (95% CI 0.35-0.86; p = 0.008; fig. 2b). The estimated NNT after adjustment was 5 (95% CI 3-19).
Calibration, internal validation and discrimination of the model were tested to address its performance. The Cox proportional-hazards regression model of 30-day mortality was well calibrated (p = 0.137). The internal validation of this model with 500-bootstrap resampling showed a reduction of <10% on the Harrell c-statistic (from 0.673 before the test to 0.655), suggesting the stability of the model.
Long-Term Mortality
The use of PAC was also associated with decreased long-term mortality in the univariate analysis (crude HR = 0.57, 95% CI 0.37 - 0.86, p = 0.007). Figure 3a shows the Kaplan-Meier survival curve. Final follow-up examinations were conducted in January 2015. The median follow-up for patients discharged from the hospital was 63 months (interquartile range: 4-83 months).
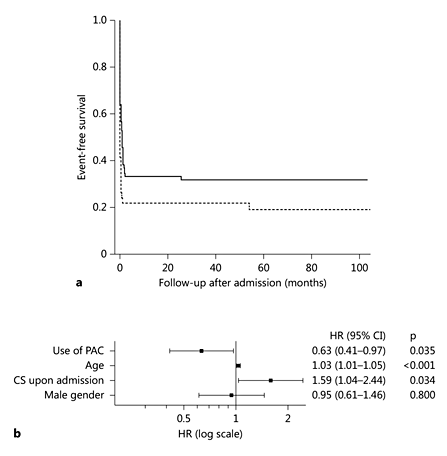
Fig. 3
Impact of PAC on long-term follow-up. a Kaplan-Meier survival estimates at follow-up. The solid line indicates the PAC group and the dashed line indicates the non-PAC group. b HR and 95% CI estimates of variables were included in the adjusted multivariate model to assess long-term mortality.
Multivariate analysis confirmed that PAC monitoring adjusted for age, gender and the presence of CS upon admission was an independent predictor for higher survival rates with an adjusted HR of 0.63 (95% CI 0.41-0.97; p = 0.035; fig. 3b). NNT after adjustment was 7 (95% uncertainty range 4-100). This model was also well calibrated (p = 0.318). The Harrell c-statistic after 500-bootstrap resampling suggested the stability of the model (from 0.659 before the test to 0.642).
Secondary End Point
Mortality at day 30 and subsequent CS was 67% in patients with ACS compared to in non-ACS patients (60%, p = 0.396). PAC did not have an impact on the mortality in patients with ACS (63% vs. 75% in non-PAC patients; p = 0.303). However, the use of PAC was associated with lower mortality in non-ACS patients (49% PAC vs. 82% in non-PAC; p = 0.010; fig. 4). Moreover, this association remained significant when it was added to our short- and long-term models adjusted by age, gender and CS upon admission. Hence, the adjusted HR of patients with PAC and non-ACS was 0.36 (95% CI 0.19-0.68, p = 0.002) and 0.48 (95% CI 0.26-0.87, p = 0.015) in the short- and long-term models, respectively.
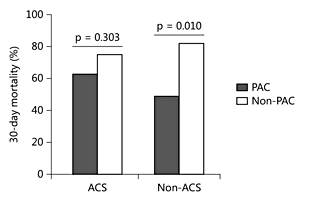
Fig. 4
Impact of PAC on 30-day mortality according to causes of CS.
Mechanical complications of acute myocardial infarction showed nonsignificant differences between groups: ventricular septal rupture (2% in non-PAC vs. 1% in the PAC group, p = 0.602) and cardiac free wall rupture (0% in non-PAC vs. 5% in the PAC group, p = 0.155). There were no papillary muscle ruptures.
Location of Venous Access and Complications Related to PAC
The location of venous access to insert the pulmonary artery catheter was mainly via the basilic vein (44%) and jugular vein (36%), with less common access gained via the femoral vein (8%) and subclavian vein (8%). Some patients were accessed via both basilic and jugular veins (4%). No deaths related to PAC complications were reported; however, 4 patients (5%) presented with major complications directly attributable to the use of PAC: atrioventricular block, ventricular tachycardia, pneumothorax and catheter-related bloodstream infection.
Discussion
After adjustment for age, gender and the presence of CS upon admission, the use of PAC in patients with CS for any cause was associated with significantly reduced short- and long-term mortality. This difference was only significant in non-ACS patients.
To our knowledge, this is the first study primarily focused upon the assessment of PAC in patients with CS. Previous studies on PAC reported small numbers of CS patients within their populations. Richard et al. [] reported 4.7% CS patients, whilst Rhodes et al. [ ]reported CS in <25% of their population. In addition, patients with CS were largely excluded in the ESCAPE (Evaluation Study of Congestive Heart Failure and Pulmonary Artery Catheterization Effectiveness) trial []. In contrast, our population represents a homogeneous sample of real-world patients with CS. The prevalence of PAC use in our study was higher than that published previously. In an assessment of trends on PAC use, Koo et al. [] showed the use of PAC in 45% of CS patients compared to the 64% in our registry.
Baseline clinical characteristics were comparable between the 2 cohorts. However, patients that underwent PAC were treated more aggressively during the hospitalization, showing a significantly higher use of inotropes as well as a nonsignificant trend for greater use of an intra-aortic balloon pump and orotracheal intubation. The use of PAC might have had an important role in the subsequent management of the patients. Unfortunately, changes in drug use and fluid management as a direct consequence of the PAC monitoring were not collected systematically. In any case, the use of inotropes has been related to higher mortality rates and the impact of the intra-aortic balloon pump on survival is nowadays under great debate.
Our main finding is that PAC is an independent predictor of survival after adjustment for age, gender and the presence of CS upon admission, showing an NNT of 5. In this regard, the use of PAC in critically ill patients has presented an historically passionate controversy. Supporters defend the use of PAC as a tool to be used in appropriately selected patients and by physicians trained to interpret and apply the data correctly, whilst their opponents suggest that clinicians are unable to correctly interpret the data obtained from PAC and even the accuracy of PAC measurements is controversial [,].
In our opinion, the use of PAC failed to show benefits in previous studies for 3 main reasons. Firstly, previous studies reported on heterogeneous populations. Beyond the assessment of patients with different degrees of heart failure, the impact of PAC on mortality has never truly been tested in a specific cohort of patients with CS. For instance, the PAC-Man study [], the largest RCT to date investigating the clinical and cost effectiveness of PAC failed to demonstrate beneficial outcomes, but the cohort comprised general ICU patients, and CS patients were not thoroughly investigated. Secondly, most trials have tested a goal-oriented hemodynamic therapy [], failing to show benefits. Noteworthy is that it remains unknown if the hemodynamic targets are appropriate surrogates, and if a particular change in therapy in response to the information provided by PAC may have an impact on mortality []. Thirdly, in addition to this complex picture, it is important to point out a probable selection bias in most previous studies, given that PAC is more likely to be used in academic hospitals and in sicker patients who are also more likely to die [,]. Altogether, our results did not contradict what has been published to date, but do shed light on the usefulness of PAC in a particular, narrowed spectrum of acute heart failure patients.
We reported an overall 30-day mortality rate of 64% in accordance with contemporary registries [,] such as in the SHOCK trial [], where mortality represented around 60% of patients. However, current CardShock study [] data show an inhospital mortality of 37%. Such a large difference may be explained because the underlying study population is different: ACS was present in 49% of our patients whereas 81% of the patients in the CardShock registry presented with ACS. In recent decades, the increase of primary percutaneous coronary intervention improved the prognosis for ACS patients, but patients without ACS did not benefit from a similar landmark therapy. Patients without ACS also have less straightforward management.
PAC use remained an independent predictor of long-term survival after adjustment, along with age and the presence of CS upon admission. However, long-term follow-up curves in both groups were parallel for 2 reasons. Firstly, CS is a serious disorder with high early mortality, but initial survivors can expect a relatively good long-term prognosis []. Secondly, the main impact of PAC upon mortality occurs during the acute phase of CS, when PAC is being used as a diagnostic tool to guide the therapy. Thus, it seems reasonable that the parameters associated with reduced short-term mortality were also associated with long-term benefits.
Our subgroup analysis based on the causes of CS revealed that PAC use was only associated with an improved short- and long-term mortality in patients without ACS. This heterogeneous group is composed mainly of patients with cardiomyopathies and valvular and pericardial diseases, and CS diagnosis in this setting is often clinically challenging. The clinical course of these patients is unpredictable, demonstrating an important role for PAC monitoring and subsequent clinical management based on these findings. In contrast, patients with ACS have a more predictable progression, making PAC less advantageous.
Major complications directly related to pulmonary artery catheter implantation were observed in 5% of patients, comparable to other studies which reported complications in 10% [] and 2.3% []. In our cohort, some complications might have been avoided due to the high proportion of peripheral venous access. Despite presenting complications, no deaths were directly attributable to insertion of the catheter. In their respective RCT, Rhodes et al. [] and Richard et al. [] also reported no mortality attributable to PAC.
Sophisticated and less invasive diagnostic and monitoring technologies are becoming increasingly available, although there is no good evidence that any form of less invasive monitoring, or even echocardiography, improves outcomes []. Echocardiography has developed a ubiquitous role in both cardiovascular disease and critical care. This technique can safely and rapidly assess cardiac structure and function, left-ventricular filling pressure and pulmonary artery pressure as well as provide accurate information regarding intravascular volume status. Thus, the ability to diagnose CS using the echocardiography-based criteria might be considered for an earlier diagnosis and improved patient safety.
Study Strengths and Limitations
The observational nature of the data does not allow demonstration of causation between the dependent variables and the outcome of PAC, stressing the need for prospective validation of our findings in a randomized trial. Despite the study being subjected to the limitations inherent to an observational registry, RCT in this setting are historically limited by methodological problems, including selection bias, noncompliance by physicians and a crossover from standard care (without PAC) to using PAC as well as to decide which PAC-guided therapeutic protocol should be used. Therefore, observational studies can provide useful information about PAC and complement that obtained by means of RCT []. Thus, although PAC was not randomly allocated, our data illustrate clinical practice in the real world.
According to Forrester et al. [] and Nohria et al. [], 4 hemodynamic and subsequent clinical patient profiles may be defined based on the presence of congestion and the adequacy of perfusion. Given that our study population was defined by the presence of CS, we assumed that our patients fulfilled the wet-cold criteria profile. However, hemodynamic and clinical data were not recorded systemically. Moreover, we lack information regarding the duration and dose of potential subsequent changes of therapy (i.e. inotropes) as a consequence of PAC, therefore limiting our conclusions on its impact on patient management.
This is a single-center study and the generalizability of our findings to other populations remains to be identified. However, this lack of external validity is compensated by the homogeneity of the sample. Complete follow-up of patients enhanced the internal validity of our results. Furthermore, the bootstrapping internal validation showed the stability of our model.
Finally, the limited number of patients and events did not allow the addition of further covariates into the multivariate adjustment without overfitting the model. Thus, larger studies are needed to confirm our results.
Conclusions
In this observational study of CS patients, PAC was associated with higher short- and long-term survival rates after adjustment for age, gender and CS upon admission. PAC use was only associated with lower mortality rates in patients without ACS, not those with ACS. Further RCT as well as larger observational studies are needed to elucidate the impact of PAC on patients with CS.
Acknowledgments
We are indebted to our teacher, Jose María Domínguez de Rozas. We also thank María Jesús Gómez Palomar for her contribution collecting data and Andrew Hall (The Hatter Cardiovascular Institute) for providing the English editing.
Conflict of Interest
There were no conflicts of interest declared.
References
- 1. Swan HJ, Ganz W, Forrester J, Marcus H, Diamond G, Chonette D: Catheterization of the heart in man with use of a flow-directed balloon-tipped catheter. N Engl J Med 1970;283:447-451.
- 2. Chatterjee K: The Swan-Ganz catheters: past, present, and future. A viewpoint. Circulation 2009;119:147-152.
- 3. Connors AF, Speroff T, Dawson N V, Thomas C, Harrell FE, Wagner D, et al: The effectiveness of right heart catheterization in the initial care of critically ill patients. SUPPORT Investigators. JAMA 1996;276:889-897.
- 4. Zion MM, Balkin J, Rosenmann D, Goldbourt U, Reicher-Reiss H, Kaplinsky E, et al: Use of pulmonary artery catheters in patients with acute myocardial infarction. Analysis of experience in 5,841 patients in the SPRINT Registry. SPRINT Study Group. Chest 1990;98:1331-1335.
- 5. Harvey S, Harrison D, Singer M, Ashcroft J, Jones CM, Elbourne D, et al: Assessment of the clinical effectiveness of pulmonary artery catheters in management of patients in intensive care (PAC-Man): a randomised controlled trial. Lancet 2005;366:472-477.
- 6. Rhodes A, Cusack RJ, Newman PJ, Grounds RM, Bennett ED: A randomised, controlled trial of the pulmonary artery catheter in critically ill patients. Intensive Care Med 2002;28:256-264.
- 7. Richard C, Warszawski J, Anguel N, Deye N, Combes A, Barnoud D, et al: Early use of the pulmonary artery catheter and outcomes in patients with shock and acute respiratory distress syndrome: a randomized controlled trial. JAMA 2003;290:2713-2720.
- 8. Binanay C, Califf RM, Hasselblad V, O'Connor CM, Shah MR, Sopko G, et al: Evaluation study of congestive heart failure and pulmonary artery catheterization effectiveness: the ESCAPE trial. JAMA 2005;294:1625-1633.
- 9. Sandham JD, Hull RD, Brant RF, Knox L, Pineo GF, Doig CJ, et al: A randomized, controlled trial of the use of pulmonary-artery catheters in high-risk surgical patients. N Engl J Med 2003;348:5-14.
- 10. Shah MR, Hasselblad V, Stevenson LW, Binanay C, O'Connor CM, Sopko G, et al: Impact of the pulmonary artery catheter in critically ill patients: meta-analysis of randomized clinical trials. JAMA 2005;294:1664-1670.
- 11. Koo KKY, Sun JCJ, Zhou Q, Guyatt G, Cook DJ, Walter SD, et al: Pulmonary artery catheters: evolving rates and reasons for use. Crit Care Med 2011;39:1613-1618.
- 12. Sotomi Y, Sato N, Kajimoto K, Sakata Y, Mizuno M, Minami Y, et al: Impact of pulmonary artery catheter on outcome in patients with acute heart failure syndromes with hypotension or receiving inotropes: from the ATTEND Registry. Int J Cardiol 2014;172:165-172.
- 13. McMurray JJ V, Adamopoulos S, Anker SD, Auricchio A, Böhm M, Dickstein K, et al: ESC guidelines for the diagnosis and treatment of acute and chronic heart failure 2012: the Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2012 of the European Society of Cardiology. Developed in collaboration with the Heart. Eur Heart J 2012;33:1787-1847.
- 14. Guidelines for treatment of acute heart failure (JCS 2011). Circ J 2013;77:2157-2201.
- 15. Yancy CW, Jessup M, Bozkurt B, Butler J, Casey DE, Drazner MH, et al: 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 2013;62:e147-e239.
- 16. Thygesen K, Alpert JS, White HD: Universal definition of myocardial infarction. Eur Heart J 2007;28:2525-2538.
- 17. Lindholm MG, Køber L, Boesgaard S, Torp-Pedersen C, Aldershvile J: Cardiogenic shock complicating acute myocardial infarction; prognostic impact of early and late shock development. Eur Heart J 2003;24:258-265.
- 18. von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP: The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Ann Intern Med 2007;147:573-577.
- 19. Vincent J-L, Pinsky MR, Sprung CL, Levy M, Marini JJ, Payen D, et al: The pulmonary artery catheter: in medio virtus. Crit Care Med 2008;36:3093-3096.
- 20. Pinsky MR, Vincent J-L: Let us use the pulmonary artery catheter correctly and only when we need it. Crit Care Med 2005;33:1119-1122.
- 21. Gattinoni L, Brazzi L, Pelosi P, Latini R, Tognoni G, Pesenti A, et al: A trial of goal-oriented hemodynamic therapy in critically ill patients. SvO2 Collaborative Group. N Engl J Med 1995;333:1025-1032.
- 22. Goldberg RJ, Samad NA, Yarzebski J, Gurwitz J, Bigelow C, Gore JM: Temporal trends in cardiogenic shock complicating acute myocardial infarction. N Engl J Med 1999;340:11621168.
- 23. Hochman JS, Sleeper LA, Webb JG, Sanborn TA, White HD, Talley JD, et al: Early revascularization in acute myocardial infarction complicated by cardiogenic shock. SHOCK Investigators. Should we emergently revascularize occluded coronaries for cardiogenic shock. N Engl J Med 1999;341:625-634.
- 24. Harjola V-P, Lassus J, Sionis A, Køber L, Tarvasmäki T, Spinar J, et al: Clinical picture and risk prediction of short-term mortality in cardiogenic shock. Eur J Heart Fail 2015;17:501-509.
- 25. Reynolds HR, Hochman JS: Cardiogenic shock: current concepts and improving outcomes. Circulation 2008;117:686-697.
- 26. Molnár Z, Vincent JL: Still a (valuable) place for the pulmonary artery catheter. Int J Cardiol 2014;173:131-132.
- 27. Forrester JS, Diamond G, Chatterjee K, Swan HJ: Medical therapy of acute myocardial infarction by application of hemodynamic subsets (second of two parts). N Engl J Med 1976;295:1404-1413.
- 28. Nohria A, Tsang SW, Fang JC, Lewis EF, Jarcho JA, Mudge GH, et al: Clinical assessment identifies hemodynamic profiles that predict outcomes in patients admitted with heart failure. J Am Coll Cardiol 2003;41:1797-1804.