Introduction
Patent foramen ovale (PFO), a congenital right-to-left interatrial shunt, is strongly associated with cryptogenic cerebrovascular events, and it has been found to be present in up to 50% of patients under 55 years old that have had a cryptogenic stroke [-]. Paradoxical embolism due to a PFO is a well-known cause of cerebrovascular events [, ]. PFO closure eliminates the shunt and has been considered the new gold standard therapy for PFO [-], especially in patients with a large shunt [-]; this highlights the importance of PFO shunt physiology in causing strokes [, , ].
In clinical practice, a residual shunt (RS) may be present in up to 25% of patients after a PFO closure, with 10% being moderate or large and having unclear significance [, , ]. Several studies have reported that RSs are a risk factor for recurrent cerebrovascular events [, , ], whereas other studies suggest otherwise [-]. In order to examine the long-term side effects of an RS in patients who have suffered a cryptogenic embolism (stroke, transient ischemic attack, or peripheral embolism) after a PFO closure, we performed a systematic review and meta-analysis of the available literature to investigate the relative effects of RSs on recurrent cerebrovascular events.
Methods and Materials
Literature Search Strategy
Two investigators independently searched the PubMed and Embase online database for relevant articles published between January 2000 and July 2021, using the following key terms: PFO; RS; right-to-left shunt; residual interatrial shunt; percutaneous PFO closure; cryptogenic embolism; cerebrovascular events; cryptogenic stroke; and cerebrovascular events’ recurrence. An additional manual search of secondary sources was also undertaken.
Inclusion and Exclusion Criteria
All original studies, including prospective or retrospective cohort studies, case series, and comparative studies, were considered. The observational studies that examined the effect of RSs on recurrent cerebrovascular events and those that reported outcome data in regard to RSs were included. Studies that were incomplete and involved fewer than 100 patients were excluded. Unpublished research was not considered.
Endpoint
The outcome was a composite ischemic endpoint comprising stroke and transient ischemic attack. Nonfatal and fatal ischemic strokes were called “stroke” in endpoint definitions across studies.
Study Quality Assessment and Data Extraction
The two reviewers independently assessed the quality of each study using the Newcastle-Ottawa Scale assessment system and guidelines about cross-section study from the Agency for Healthcare Research and Quality. Information collected included the principal author, year of publication, study design, sample size, recurrence, and methods for RS detection, such as transesophageal echocardiography, contrast transesophageal echocardiography, transthoracic echocardiography, and contrast transthoracic echocardiography cTTE. Basic clinical data, such as average age, proportion of male/female, and postprocedure medication, were also collected.
Statistical Analysis
Dichotomous data were presented as odds ratios (ORs) and corresponding 95% confidence intervals (CIs), using random- or fixed-effect models. ORs and 95% CIs were used to present the association between RSs and recurrent cerebrovascular events, and the distribution was represented using a forest plot. When statistical pooling was required, the random-effect model was used for heterogeneity; otherwise, the fixed-effects model was used []. The I2 statistic was calculated to demonstrate the degree of heterogeneity, specifically, and the percentage of variation across studies that was not due to chance. Heterogeneity was considered low if I2 < 25%, moderate if I2 was 25–50%, and significant if I2 > 50%. Publication bias was assessed graphically using the funnel plot and further assessed using an Egger’s test. The analysis was undertaken using the Stata 15 software.
Results
A total of 2,083 patients who had experienced a prior cerebrovascular event related to PFO and who met the inclusion criteria were enrolled in six studies [-] (Fig. 1). The study quality was assessed using the Newcastle-Ottawa Scale and cross-section study guidelines from the Agency for Healthcare Research and Quality; both were rated a total score of ≥6. The full characteristics of each study are detailed in Table 1.
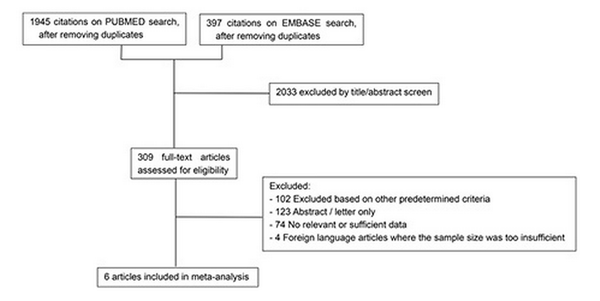
Fig. 1
Flow diagram demonstrating the search results. Six reports were included in the meta-analysis.
RSs induced a significant increase in recurrent cerebrovascular events (OR = 3.484; 95% CI, 2.169–5.596) (Fig. 2) with low heterogeneity (I2 = 0.0%). Across all the studies, 32 out of 360 patients had a recurrence of cerebrovascular events in the RS arms, compared with 50 out of 1,723 in other arms. Overall, the annual risk for recurrent cerebrovascular events was lower in the non-RS group (2.90%) than in the RS group (8.89%).

Fig. 2
Forest plot of OR for recurrent cerebrovascular events in RSs and non-RSs.
Test for Publication Bias
Figure 3 depicts a funnel plot for the six studies of recurrent cerebrovascular events after percutaneous PFO closure. The funnel plot demonstrates asymmetry, which suggests possible publication bias. The results of the Egger’s test suggested a statistically significant asymmetric distribution of the six studies (p = 0.536; 95% CI, 1.619–2.661).
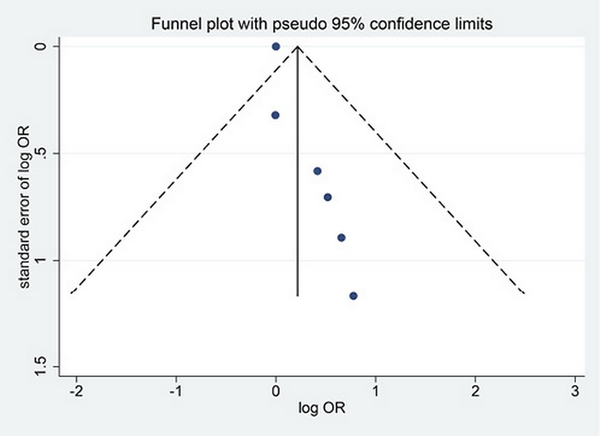
Fig. 3
Funnel plot for publication bias.
Discussion
The association between recurrent cerebrovascular events with or without RSs in adults who had cerebrovascular events after PFO has been confirmed, as shown in Figure 2. It has been postulated that PFO closure prevents cerebrovascular events because of the passage of paradoxical embolisms through the PFO shunt into the arterial circulation. As a consequence, serotonin, homocysteine, and other vasoactive factors that are normally metabolized in the pulmonary circulation enter the cerebral vasculature, which can provoke attacks of thrombosis.
It would appear that the presence of an RS may increase concentrations of serotonin and homocysteine signaling agents on the arterial side or provide the passage of paradoxical embolisms into the arterial circulation after successful PFO closure to prevent the secondary cerebrovascular events. In this meta-analysis, it was determined that such explanations could account for the increase in recurrent cerebrovascular event attacks of patients with an RS after PFO closure.
In clinical practice, the aim of limiting recurrent events to the first 2 years after percutaneous PFO closure is of note, especially in the first year. Only a few studies [-] suggested limited specific predictors of recurrent cerebrovascular events after PFO closure, including the activity of the primary septum and atrial septal aneurysm, which were needed for large-scale clinical trials to demonstrate. The Randomized Evaluation of Recurrent Stroke Comparing PFO Closure to Established Current Standard of Care Treatment trial [] found a higher risk of stroke recurrence only in patients with a larger right-to-left shunt. The results of this meta-analysis suggest that RSs may be a risk factor for recurrent cerebrovascular events. However, the analysis did not look at the risk of different shunt sizes for two reasons. First, RS size diminishes over time, usually within 3 months after surgery, as the closure device becomes further epithelialized; the time varies for every patient. Second, the assessment methods and classification of RSs in this meta-analysis were not uniform, meaning that the classification of size was not precise.
Limitations
There are several limitations in the present meta-analysis worth mentioning when drawing conclusions regarding the risks of an RS in patients who have experienced recurrent cerebrovascular events. First, the small size of the included literature and the limited number of patients, combined with a recall bias cannot be excluded, considering that most of the included studies were retrospective. Second, the baseline characteristics and postsurgical protocol for assessing the outcomes might differ between studies, resulting in some imprecision of the results due to heterogeneity. Third, as noted above, the surgical procedures employed several different devices. Finally, according to the researchers in question, the baseline data on sex and age were not recorded in three of the studies.
Conclusions
This systematic literature review and meta-analysis has confirmed that the presence of an RS serves as a risk factor for recurrent cerebrovascular events in postsurgical patients who had PFO-related cerebrovascular events. Therefore, the risk of RSs in patients at high risk of recurrent cerebrovascular events should also not be ignored in the clinic. Due to the possibility of bias arising from this retrospective analysis, future research in this area is needed to demonstrate conclusively the prognostic value of RSs after PFO closure for recurrent cerebrovascular event outcomes.
Acknowledgments
We are particularly grateful to all the people who have given us help on our article.
Statement of Ethics
An ethics statement is not applicable because this study is based exclusively on published literature. All participants signed a document of written informed consent.
Conflict of Interest Statement
The authors declare that they have no competing interests.
Funding Sources
No external funding received to conduct this study.
Authors Contributions
Conception and design of the research: Rong-Hong Jiao, Zhi-An Jang, and Ting-Ting Liu. Acquisition of data: Ting-Ting Liu and Tao Chen. Analysis and interpretation of the data: Ting-Ting Liu, Tao Chen, and Wen-Lou Bai. Statistical analysis: Ting-Ting Liu, Tao Chen, and Wen-Lou Bai. Obtaining financing: none. Writing of the manuscript: Ting-Ting Liu. Critical revision of the manuscript for intellectual content: Ting-Ting Liu and Tao Chen. All authors read and approved the final draft.
Data Availability Statement
All data generated or analyzed during this study are included in this article. Further enquiries can be directed to the corresponding author.
References
- 1. Kent DM, Dahabreh IJ, Ruthazer R, Furlan AJ, Reisman M, Carroll JD, et al. Device closure of patent foramen ovale after stroke: pooled analysis of completed randomized trials. J Am Coll Cardiol. 2016;67(8):907–17. https://doi.org/10.1016/j.jacc.2015.12.023.
- 2. Handke M, Harloff A, Olschewski M, Hetzel A, Geibel A. Patent foramen ovale and cryptogenic stroke in older patients. N Engl J Med. 2007;357(22):2262–8. https://doi.org/10.1056/nejmoa071422.
- 3. Abdelghani M, El-Shedoudy SAO, Nassif M, Bouma BJ, de Winter RJ. Management of patients with patent foramen ovale and cryptogenic stroke: an update. Cardiology. 2019;143(1–2):62–72. https://doi.org/10.1159/000501028.
- 4. Ropper AH. Tipping point for patent foramen ovale closure. N Engl J Med. 2017;377(11):1093–5. https://doi.org/10.1056/nejme1709637.
- 5. Meier B, Kalesan B, Mattle HP, Khattab AA, Hildick-Smith D, Dudek D, et al. Percutaneous closure of patent foramen ovale in cryptogenic embolism. N Engl J Med. 2013;368(12):1083–91. https://doi.org/10.1056/nejmoa1211716.
- 6. Alkhouli M, Sievert H, Holmes DR. Patent foramen ovale closure for secondary stroke prevention. Eur Heart J. 2019;40(28):2339–50. https://doi.org/10.1093/eurheartj/ehz157.
- 7. Kuijpers T, Spencer FA, Siemieniuk RAC, Vandvik PO, Otto CM, Lytvyn L, et al. Patent foramen ovale closure, antiplatelet therapy or anticoagulation therapy alone for management of cryptogenic stroke? A clinical practice guideline. BMJ. 2018;362:k2515. https://doi.org/10.1136/bmj.k2515.
- 8. Saver JL, Carroll JD, Thaler DE, Smalling RW, MacDonald LA, Marks DS, et al. Long-term outcomes of patent foramen ovale closure or medical therapy after stroke. N Engl J Med. 2017;377(11):1022–32. https://doi.org/10.1056/nejmoa1610057.
- 9. Mas JL, Derumeaux G, Guillon B, Massardier E, Hosseini H, Mechtouff L, et al. Patent foramen ovale closure or anticoagulation vs. antiplatelets after stroke. N Engl J Med. 2017;377(11):1011–21. https://doi.org/10.1056/nejmoa1705915.
- 10. Søndergaard L, Kasner SE, Rhodes JF, Andersen G, Iversen HK, Nielsen-Kudsk JE, et al. Patent foramen ovale closure or antiplatelet therapy for cryptogenic stroke. N Engl J Med. 2017;377(11):1033–42. Corrected and republished from: N Engl J Med. 2020 Mar 5;382(10):978. https://doi.org/10.1056/nejmoa1707404.
- 11. Chen X, Chen SD, Dong Y, Dong Q. Patent foramen ovale closure for patients with cryptogenic stroke: a systematic review and comprehensive meta-analysis of 5 randomized controlled trials and 14 observational studies. CNS Neurosci Ther. 2018;24(10):853–62. https://doi.org/10.1111/cns.12980.
- 12. Windecker S, Wahl A, Nedeltchev K, Arnold M, Schwerzmann M, Seiler C, et al. Comparison of medical treatment with percutaneous closure of patent foramen ovale in patients with cryptogenic stroke. J Am Coll Cardiol. 2004;44(4):750–8. https://doi.org/10.1016/j.jacc.2004.05.044.
- 13. Serena J, Segura T, Perez-Ayuso MJ, Bassaganyas J, Molins A, Dávalos A. The need to quantify right-to-left shunt in acute ischemic stroke: a case-control study. Stroke. 1998;29(7):1322–8. https://doi.org/10.1161/01.str.29.7.1322.
- 14. Overell JR, Lees KR, Bone I. Percutaneous closure of patent foramen ovale in patients with paradoxical embolism. Circulation. 2001;103(10):E56. https://doi.org/10.1161/01.cir.103.10.e56.
- 15. Spies C, Strasheim R, Timmermanns I, Schraeder R. Patent foramen ovale closure in patients with cryptogenic thrombo-embolic events using the Cardia PFO occluder. Eur Heart J. 2006;27(3):365–71. https://doi.org/10.1093/eurheartj/ehi617.
- 16. Caputi L, Butera G, Anzola GP, Carminati M, Carriero MR, Chessa M, et al. Residual shunt after patent foramen ovale closure: preliminary results from Italian patent foramen ovale survey. J Stroke Cerebrovasc Dis. 2013;22(7):e219–26. https://doi.org/10.1016/j.jstrokecerebrovasdis.2012.12.002.
- 17. Agarwal S, Bajaj NS, Kumbhani DJ, Tuzcu EM, Kapadia SR. Meta-analysis of transcatheter closure versus medical therapy for patent foramen ovale in prevention of recurrent neurological events after presumed paradoxical embolism. JACC Cardiovasc Interv. 2012;5(7):777–89. https://doi.org/10.1016/j.jcin.2012.02.021.
- 18. Wallenborn J, Bertog SC, Franke J, Steinberg DH, Majunke N, Wilson N, et al. Recurrent events after percutaneous closure of patent foramen ovale. Catheter Cardiovasc Interv. 2013;82(4):541–6. https://doi.org/10.1002/ccd.24511.
- 19. Whitehead A, Whitehead J. A general parametric approach to the meta-analysis of randomized clinical trials. Stat Med. 1991;10(11):1665–77. https://doi.org/10.1002/sim.4780101105.
- 20. Kiblawi FM, Sommer RJ, Levchuck SG. Transcatheter closure of patent foramen ovale in older adults. Catheter Cardiovasc Interv. 2006;68(1):136–42. https://doi.org/10.1002/ccd.20722.
- 21. Sievert H, Horvath K, Zadan E, Krumsdorf U, Fach A, Merle H, et al. Patent foramen ovale closure in patients with transient ischemia attack/stroke. J Interv Cardiol. 2001;14(2):261–6. https://doi.org/10.1111/j.1540-8183.2001.tb00745.x.
- 22. Wahl A, Meier B, Haxel B, Nedeltchev K, Arnold M, Eicher E, et al. Prognosis after percutaneous closure of patent foramen ovale for paradoxical embolism. Neurology. 2001;57(7):1330–2. https://doi.org/10.1212/wnl.57.7.1330.
- 23. Karagianni A, Mandalenakis Z, Dellborg M, Mirzada N, Johansson MC, Eriksson P. Recurrent cerebrovascular events in patients after percutaneous closure of patent foramen ovale. J Stroke Cerebrovasc Dis. 2020;29(8):104860. https://doi.org/10.1016/j.jstrokecerebrovasdis.2020.104860.
- 24. Hammerstingl C, Bauriedel G, Stüsser C, Momcilovic D, Tuleta I, Nickenig G. Risk and fate of residual interatrial shunting after transcatheter closure of patent foramen ovale: a long term follow up study. Eur J Med Res. 2011;16(1):13–9. https://doi.org/10.1186/2047-783x-16-1-13.
- 25. Deng W, Yin S, McMullin D, Inglessis-Azuaje I, Elmariah S, Hung J, et al. Residual shunt after patent foramen ovale closure and long-term stroke recurrence. Ann Intern Med. 2020;173(11):946–7. https://doi.org/10.7326/l20-1274.
- 26. Lee PH, Song JK, Kim JS, Heo R, Lee S, Kim DH, et al. Cryptogenic stroke and high-risk patent foramen ovale: the DEFENSE-PFO trial. J Am Coll Cardiol. 2018;71(20):2335–42. https://doi.org/10.1016/j.jacc.2018.02.046.
- 27. Mas JL, Arquizan C, Lamy C, Zuber M, Cabanes L, Derumeaux G, et al. Recurrent cerebrovascular events associated with patent foramen ovale, atrial septal aneurysm, or both. N Engl J Med. 2001;345(24):1740–6. https://doi.org/10.1056/nejmoa011503.
- 28. Thaler DE, Ruthazer R, Weimar C, Mas JL, Serena J, Di Angelantonio E, et al. Recurrent stroke predictors differ in medically treated patients with pathogenic vs. other PFOs. Neurology. 2014;83(3):221–6. https://doi.org/10.1212/wnl.0000000000000589.
- 29. Carroll JD, Saver JL, Thaler DE, Smalling RW, Berry S, MacDonald LA, et al. Closure of patent foramen ovale versus medical therapy after cryptogenic stroke. N Engl J Med. 2013;368(12):1092–100. https://doi.org/10.1056/nejmoa1301440.