Introduction
In the practice of clinical pharmacology, there is probably no concept more contentious or misunderstood than that of Narrow Therapeutic Index (NTI) Drugs. In theory, it is a simple concept that there exists in medicine for which both dose titration and routine clinical monitoring are essential for effective therapy because the dose associated with toxicity is not much greater than the dose required for efficacy. The identification of such drugs and the call for stricter regulation has made the issue a touchstone for the clinician, patient and regulator. The controversy surrounding the approval of generic warfarin (Coumadin), generic substitutions, and the activity of special interest groups who harness the Narrow Therapeutic Index classification for their benefit readily come to mind when one looks at this issue.[] Coumadin (warfarin) first came into commercial use in 1948 as rat poison. However, in 1954 it was approved for medical use in the USA.
The scientific community has been clearly divided. While some have supported[] the FDA position[] on the general issue of generic substitution or the approval of generic warfarin, others either opposed or were reluctantly in support until more convincing clinical data became available.[] To our knowledge, to date there are convincing clinical data that have been reported in the literature to show that the use of generic warfarin has been associated with any potential risk.[, , , , , , , , , ]
In response to various elements contained in this debate, the Food and Drug Administration (FDA) has had cause to evaluate its current practices and to propose, where appropriate, modification to its various guidelines and guidances. One of these initiatives was assessment of the way Narrow Therapeutic Index drugs were identified and how they were regulated. As part of the process, the current (circa 1970s) regulatory definition of a Narrow Therapeutic Index drug was found to be inadequate for either a researcher, clinician or regulator. The objective of this commentary is to lay out the framework for the development of a new regulatory definition of a Narrow Therapeutic Index drug that is both practical and clinically relevant.
The Food and Drug Administration (FDA) ‘Office of Generic Drugs | FDA’ and The European Medicines Agency (Science Medicines Health) are located in the following link ‘Product-specific bioequivalence guidance | European Medicines Agency (europa.eu)’ provided a plethora of information, guidance and guidelines for the development of generic NTI drugs as well as and new drugs ‘Center for Drug Evaluation and Research | CDER | FDA’. Also, the FDA published a list of NTI in November 1995 in the Guidance for Industry for Immediate Release Solid Oral Dosage From and better known as SUPAC-IR (Supplementary Appendix 1; https://www.fda.gov/regulatory-information/search-fda-guidance-documents/supac-ir-immediate-release-solid-oral-dosage-forms-scale-and-post-approval-changes-chemistry).
For more details, the readers should also consult the FDA, Center for Drug Evaluation and Research-CDER (Center for Drug Evaluation and Research | CDER | FDA) and the Office of Generic Drugs (Generic Drugs | FDA). In addition, the European Medicines Agency-EMA at ‘https://www.ema.europa.eu/en/human-regulatory/research-development/scientific-guidelines/clinical-pharmacology-pharmacokinetics/product-specific-bioequivalence-guidance’.
Terminology
At the present time, there is no clear consensus on the terminology, definition or characteristics of what constitutes a Narrow Therapeutic Index drug. Several terms such as narrow therapeutic index (NTI), narrow therapeutic window, narrow therapeutic range and narrow therapeutic ratio, have been used interchangeably. These drugs have also been referred to as complicated drugs, critical dose drugs, critical use drugs, critical bioavailability drugs, problem drugs and difficult drugs.[]
The term narrow therapeutic index and its abbreviated term Narrow Therapeutic Index are most widely used in this area, but they themselves are subject to interpretation. While the terms ‘narrow’ and ‘therapeutic’ have achieved generally agreed to meanings, their modifier ‘index’ and ‘range’ are more often than not misunderstood. From a purely lexicographic view, Webster's Dictionary defines the term index as a ‘guide’ or a ‘list’ that can be short or long. It is not intended to describe a magnitude, such as ‘narrow or wide’. Similarly, the term ratio can be defined as ‘low or high’, but not ‘narrow or wide’. Conversely, the terms ‘range or window’ can have a magnitude modifier such as ‘width’ associated with them. From a strictly linguistic point of view, the proper terminology should be either narrow therapeutic window or range.
In an effort to evaluate the widespread nature and use of these terms, a search of various biomedical databases was conducted. The results of this literature survey are summarized in Supplementary Table 1. Clearly, although linguistically awkward, the most commonly used phraseology to describe these drugs is Narrow Therapeutic Index drugs or Narrow Therapeutic Index's. Because of its extant nature in the literature, although it may not be ‘proper’, this terminology has acquired a legitimacy in the scientific community literature that is not worth challenging.
The objective of this survey was to establish the pattern of use of these terms along with other common variations. The results of this investigation, summarized in Supplementary Table 1, indicate that both ‘Narrow Therapeutic Index’ and ‘Narrow Therapeutic Range’, and their variants, are used to almost the same extent in the literature. Because of this general acceptance of the two terms, it is apparent that their domination over other alternatives makes either of them an acceptable choice based on usage alone. Although ‘Narrow Therapeutic Range’ is the ‘proper’ term, we have elected to use ‘Narrow Therapeutic Index’ in this paper as it is both widely used and from a medical point of view, it is conceptually appealing.
Regulatory Definition of Narrow Therapeutic Index Drugs
At present, the FDA does not designate specific drugs as Narrow Therapeutic Index's. However, in the Code of Federal Regulation (21 CFR 320.33c), the FDA has provided a definition for drugs with a narrow therapeutic ratio based on median lethal and effective doses. This definition is as follows:
Evidence that the drug products exhibit a narrow therapeutic ratio, e.g., there is less than a 2-fold difference in the median lethal dose (LD50) and the median effective dose (ED50) values, or have a less than 2-fold difference in the minimum toxic concentrations (MTC) and the minimum effective concentrations (MEC) in the blood and safe and effective use of the drug products requires careful dosage titration and patient monitoring.
This definition is not especially useful to clinicians or regulators. The definition itself is flexible in that it uses both animal and human data to establish a therapeutic ratio, but these very elements are also the definition's weak points. The LD50 test is not routinely conducted today because of animal rights concerns. In addition, the determination of MTC and MEC is not a routine goal of drug development. However, it should be noted that the definition does emphasize the importance of dosage titration and patient monitoring.
The Canadian Health Protection Bureau, in a 1997 draft guidance on Narrow Therapeutic Index drugs, identified Narrow Therapeutic Index drugs[, ] as having
...a less than 2-fold difference in the ratio between the lowest concentration at which the clinical toxicity commonly occurs to the median concentration providing a therapeutic effect.
This definition is more practical as it does not rely upon LD50 values and is based on the clinical experience with the drug. Both the US and Canadian definitions are an attempt to describe a situation where the relative width of an ‘index’ determines whether or not a drug is an Narrow Therapeutic Index drug. From a clinical pharmacology perspective, this is the equivalent of measuring the difference between the concentration–therapeutic effect curve for a drug and its corresponding concentration–adverse event curve (Figure 1). Thus, the efficacy and safety of a Narrow Therapeutic Index drug can be thought of as being a function of the steepness of its dose–response relationship and the degree of separation of concentrations that elicit the therapeutic and dose-limiting adverse effects. Based on this relationship and incorporating the need for careful dose titration at clinical doses, we propose the following definition as a more clinically relevant and useable regulatory definition of a Narrow Therapeutic Index drug:
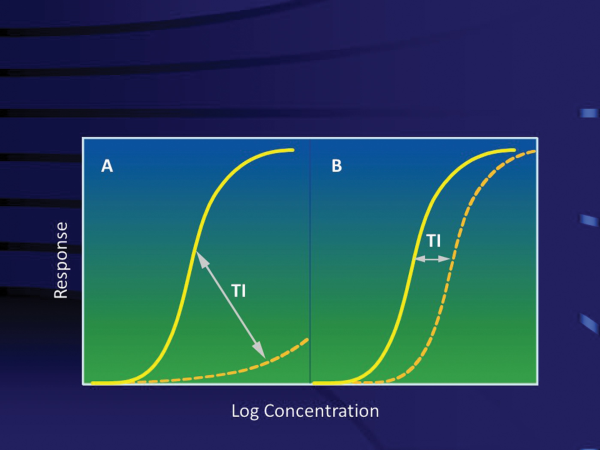
Figure 1
Dose–response relationship for wide (A) and Narrow (B) Therapeutic Index (TI) drugs (solid line = efficacy curve and dotted line = toxicity curve).
NTI drugs are those with the potential to cause dose-limiting adverse effects at or near doses required to achieve the desired therapeutic effect and which, in clinical practice, routinely require careful incremental dose titration and quantitative monitoring of drug concentrations or pharmacodynamic effects.
In the FDA guidance document entitled ‘Bioavailability and Bioequivalence Studies for Orally Administered Drug Products – General Considerations’, the following definition of a narrow therapeutic range drug is provided:
This guidance defines narrow therapeutic range drug products as those containing certain drug substances that are subject to therapeutic drug concentration or pharmacodynamic monitoring, and/or where product labeling indicates a narrow therapeutic range designation.
However, at the recent Canadian Expert Advisory Committee on Bioavailability and Bioequivalence meeting held in March 2001,[] the Narrow Therapeutic Index drugs, referred to as ‘critical drugs’ were defined as:
Comparatively small differences in dose or concentration lead to serious therapeutic failure and/or adverse drug reactions which may be persisted, irreversible, slowly reversible, or life-threatening events.
The definition in the FDA guidance is consistent with our proposed revised definition except that it refers to these drugs as having a ‘range’ instead of an ‘index’. While this is the preferred way to refer to the difference between effective and toxic blood concentrations, it is not the most commonly used and indexed term found in the literature. As such, we have chosen to use index in my definition as it relates to them and have provided my reasons for doing so above.
To summarize, it is this concept of a concentration–response curve and its delineation during drug development that we believe is at the heart of the Narrow Therapeutic Index issue. Ideally, this would allow for the identification of Narrow Therapeutic Index drugs during the drug development process.
Unfortunately, at present we do not, nor am we likely to have the information needed to map out either the effect or toxicity curves so often noted in textbooks as being the underpinnings of dose selection. Only through better dose ranging and more flexible dosing strategies in clinical trials can we hope to map out these relationships sufficiently such that we can truly provide the clinician in the label the lowest efficacious dose that maximizes efficacy and minimizes toxicity.
Variability and Utility of PK/PD Monitoring
In the proposed definition above, Narrow Therapeutic Index drugs are those that are subject to therapeutic drug monitoring (TDM) based on pharmacokinetic (PK) or pharmacodynamic (PD) measures. The need for PK or PD monitoring relates to the type of variability inherent in the PK/PD measures of these drugs, and the following principles generally apply:
To be clinically useful and predictable, Narrow Therapeutic Index drugs must have low intrasubject variability. From the literature review, it can be stated that virtually all Narrow Therapeutic Index drugs have low intrasubject variability in PK and PD measures.[, ] From an examination of available data for drugs routinely considered to be Narrow Therapeutic Index drugs from the literature review, it can be stated that virtually all Narrow Therapeutic Index drugs have low intrasubject variability in PK and PD measures. This observation is well support by others.
In contrast, the intersubject variability of PK or PD measures is relatively high for Narrow Therapeutic Index drugs and hence, therapeutic monitoring is needed for dose individualization. When the intersubject variability of a PD measure is high, PD monitoring is required for dose selection and titration. Conversely, if the intersubject variability of a PK measure is high and a PK–PD relationship exists, PK monitoring is often used for dose optimization.
Based on these observations, it is readily seen that the utility of PK or PD monitoring for a Narrow Therapeutic Index drug is associated with both intra- and intersubject variability. When PD variability is high and PK variability is low, the value of PK monitoring is not useful.[] A good example is warfarin. Monitoring of PD response (e.g. prothrombin time) of warfarin plays an important role in titration and dose selection whereas measurement of its plasma concentrations has little utility in optimizing therapy. The optimal dose of warfarin varies widely between patients because of extrinsic and intrinsic factors such as PK variability, genetics and diet.[] The high intersubject variability in the PD response necessitates PD monitoring for individualization of warfarin dose. The low intrasubject variability in the PD response to warfarin enables the prescriber to predict the clinical response within that patient. It should be noted that Coumadin (warfarin) was given to the US president Dwight David Eisenhower when he had heart attack in 1955.
In contrast, cyclosporine,[] phenytoin[, ] and procainamide[] are and exemplify drugs with a direct PK–PD relationship where the PK intersubject variability between patients is relatively high.[] Here, monitoring of plasma concentrations is critical in achieving optimal efficacy and safety, yet PD monitoring may also be important for the optimization of therapy.
There are several sources of PK and PD variability that a clinician must be aware of. In many cases, these sources combine to result in a greater overall variability in clinical responses. Understanding the sources of this variability becomes important in optimizing drug therapy.
Risk Management and Atypical Narrow Therapeutic Index Drugs
In my reviews, they focused on ‘risk management’ in terms of the relationship between the risk/benefit ratio and severity of the disease. The toxicity of a drug and severity of a disease being treated cannot be considered in isolation. The main questions that should be considered by the clinician are:
How safe and effective is the drug?
What is the disease being treated?
What is the duration of treatment?
What is the degree or severity of adverse events?
What is the duration of adverse events?
These questions are especially relevant in the clinical use of Narrow Therapeutic Index drugs. Rational drug therapy requires knowledge about the ratio of the risk of adverse drug reactions to therapeutic benefit.
As mentioned, a Narrow Therapeutic Index drug can be considered in the context of the steepness of its dose–response relationship and degree of separation between the two dose–response curves that elicit the therapeutic and adverse effects. There are some drugs with a single indication, but with multiple toxicity as shown in Figure 2. By contrast, there are some atypical Narrow Therapeutic Index drugs that have more than one indication and thus, more than one dose–response relationship and toxicity profiles, depending on the dose.
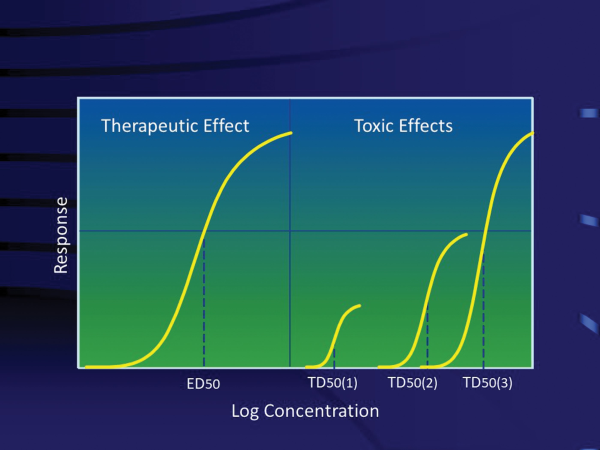
Figure 2
Drug concentration-efficacy and toxicity relationship (solid line = efficacy curve and dotted line = toxicity curve). ED50 = The dose or concentration producing 50% of maximum efficacy and TD50 = dose or concentration producing 50% of maximum toxicity.
A good example of trade-off between risk and efficacy is aspirin. The history of aspirin begins with its synthesis and manufacture in 1899. In 1915, it became available to the public as Over the Counter (OTC). Aspirin has a very wide range of doses from 60 mg a day for antiplatelet effects, to 650 mg every 4–6 h for headache, up to 4–6 g a day for rheumatoid arthritis.[] Clearly, most clinicians do not consider aspirin a Narrow Therapeutic Index drug. However, in its use as an antirheumatic drug, the dose required to achieve the therapeutic effects is close to the dose that results in undesirable toxicity.[, ]
In this situation, aspirin exhibits the characteristics of a Narrow Therapeutic Index drug.[] Aspirin typifies a drug that has multiple therapeutic uses and for each indication has a dose-limiting toxicity associated with it (Figure 3). Whether or not dose-limiting toxicity is related to a direct extension of pharmacologic effect, such as inhibition of prostaglandin production in gastritis or is due to another mechanism, such as tinnitus associated with high dose rheumatoid arthritis therapy, the potential for an adverse event to occur in a particular patient must be weighed against the anticipated therapeutic benefit to the patient. This differs from the Narrow Therapeutic Index drugs such as phenytoin and digoxin in that the latter drugs exhibit serious dose-limiting toxicities over their entire range of therapeutic doses and require for all doses either PK or PD monitoring, something that for aspirin is rarely done even at high doses. It is noteworthy that digoxin is no longer the first choice in heart failure. It may even increase the risk of death. The current recommendation of the American Stroke and Heart Association is the use of a triple therapy of angiotensin-converting enzyme (ACE) inhibitor, beta-blocker and mineralocorticoid receptor antagonist (e.g. spironolactone and eplerenone) to heart failure. Digoxin is a third-line therapy.
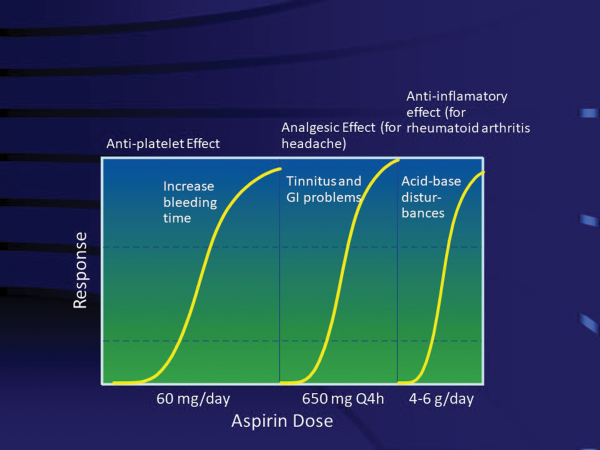
Figure 3
Dose–response relationship for aspirin as example of atypical drug with multiple indications and multiple dose–response profiles (response depends on individual patient sensitivity to aspirin).
As mentioned above, a Narrow Therapeutic Index drug can be considered in the context of the steepness of its dose–response relationship and the degree of separation between the two dose–response curves that elicit the therapeutic and adverse effects (Figure 1). Having made this argument one should realize that certain classes of drugs such as antineoplastic agents and volatile anaesthetics, having some or most of the typical characteristics described above, might be considered definitely Narrow Therapeutic Index drugs, but these are closely monitored by the anaesthetics during the surgery. These drugs, by their very nature, are dosed at levels where the distance between therapeutic and toxic concentrations is in most instances either extremely narrow or overlapping. However, because of their inherent toxicity, these drugs are used only in specialized settings by trained personnel whose knowledge and training substantially improves the risk–benefit ratio to the patient. These drugs are definitely considered as Narrow Therapeutic Index drugs. The applicability and explanation apply to the chemotherapy drugs.
Bioequivalence and Narrow Therapeutic Index Drugs
The bioequivalence methods and standards used by the FDA in comparing a generic with an innovator drug product are the same as those used to compare changes in formulations or manufacturing of innovator products. For example, the innovator usually conducts bioequivalence studies between the pivotal clinical trial formulation and the to-be-marketed drug formulation if those formulations are not the same. Following postapproval changes in production scale, equipment, manufacturing procedures, manufacturing site, inactive ingredients or dosage form, the innovator may conduct bioequivalence studies to determine if the postchange formulation is bioequivalent to the prechange formulation. In these situations, the FDA applies the same bioequivalence criteria as when evaluating a new generic drug product. When an innovator changes the formulation of a Narrow Therapeutic Index drug, the studies needed to support the change are the same as those needed for approval of generic formulators of the same Narrow Therapeutic Index drug.
There are no additional approval requirements for generic versions of Narrow Therapeutic Index drugs versus non-Narrow Therapeutic Index drugs. The Agency does not set specific standards based on a therapeutic index. The bioequivalence criteria, using the 90% confidence interval approach, are quite strict (i.e. 80–125%). In a review published in JAMA in 1999 of the 127 bioequivalence studies submitted to the Office of Generic Drugs in 1997, the mean difference in bioequivalence parameters between the approved generic and reference listed drug was less than 4.3%.[] Thus, there is no need to apply stricter criteria for Narrow Therapeutic Index drugs.
In addition, the quality controls imposed through established strict chemistry and manufacturing specifications and monitoring, as well as other product quality tests, assure that the original determination of equivalence is valid throughout the time when the products are on the market.
The current FDA position is that a generic product may be switched with its corresponding reference listed drug.[, ] In approving all generic drugs, the FDA requires rigorous tests and procedures to assure that the generic drug is interchangeable with the reference drug product under all conditions of use.
It should be noted that the regulatory Agency in Japan is in agreement with the FDA in terms of the CI limits of 80–125% for all drugs, including Narrow Therapeutic Index drugs.[] However, in Canada, at the Canadian Expert Advisory Committee on Bioavailability and Bioequivalence meeting held in March 2001, narrowing the 90% CI limits was suggested to be 90–112% for Narrow Therapeutic Index drugs.[] However, for the effect of food studies involving Narrow Therapeutic Index drugs, the 90–112% CI limit was suggested to be applied for AUC data only, but not Cmax. Furthermore, the Canadians noted that they are planning to compile a list of Narrow Therapeutic Index drugs in the future. Earlier, the Canadian Society for Pharmaceutical Sciences Discussion Group proposed the use of the 95% CI limits instead of 90% for both Cmax and AUC.[]
At the April 2010 FDA Advisory Committee meeting on Narrow Therapeutic Index, the committee voted 1–2 that the bioequivalence (BE) limit of a 90% confidence interval with 80–125% is insufficient for the Narrow Therapeutic Index drugs.[] In this report, the proposed BE limits for Narrow Therapeutic Index drugs of 90.00–111.11% would be scale based on the within-subjects variability of the reference drugs. Similarly, for cyclosporine, the authors stated that the regulatory authorities required both Cmax and AUC in Europe.[] In another report,[] drug-related problems (DRP), such as warfarin, aminoglycosides, carbamazepine and lithium, were associated with 35% DRP (292 out 827 patients).
Conclusion
One objective of drug regulation is to obtain a favourable risk/benefit ratio based on the condition being treated and the duration of treatment. In this article, we have discussed the current regulatory status of Narrow Therapeutic Index drugs in the USA and have proposed a definition based on clinical pharmacology principles. By attempting to incorporate both dose-limiting toxicity and the need for close patient monitoring and titration in our proposal, we have attempted to make the definition more useful to the clinician and regulator. The FDA has refrained from publishing a list of Narrow Therapeutic Index drugs for a variety of reasons, including but not limited to the fact that all such lists have some degree of bias in them and the fact that Narrow Therapeutic Index drugs defy grouping by simple criteria. It is the magnitude or severity of the clinical outcome as a consequence of either the drug therapeutic failure or the toxicity that defines a drug as having a narrow therapeutic index.
The current FDA position is that, regardless of whether it is a Narrow Therapeutic Index or non-Narrow Therapeutic Index drug, generic products may be switched with the corresponding reference listed drug without any additional regulatory monitoring. In approving all generic drugs, the FDA requires rigorous tests and procedures to assure that the generic drug is interchangeable with the reference drug product under all conditions of use. Furthermore, it would be very helpful to the health system if more time was spent on physician's and prescriber's education, especially on the understanding of the current FDA standards and requirements in approving generic drugs.
The NTI drugs and non-NTI drugs cannot be treated the same and generic NTI drugs shall not be switched with corresponding RLD's with only conventional average BE limits of 80.00–125% and need additional tests of comparison of test and reference product within-subject variability.
Acknowledgments
This paper was part of the team at the Food and Drug Administration (FDA) that was formed in 1997 as at the time of Generic drug application for warfarin. I would like to thank the following individuals with the FDA and outside the FDA (in random order) for their discussion throughout the years: Drs. John Balian (Former FDA, left to Pfizer), Roger Williams (Former FDA, left to USP), Thomas Tozer (University of California at San Francisco), Jurgen Venitz (Virginia Commonwealth University at Richmond), Larry Lesko (Former, FDA, Unknown), E. Dennis Bashaw (Former FDA, Unknown), Dale P. Conner (Former FDA, Unknown), Mei-Ling Chen (Former FDA, Unknown), and Barbara M. Davit (Former FDA, Unknown).
References
- 1. Habet S, Bashaw D, Lesko L, et al Narrow Therapeutic Index (NTI) Drugs. Preliminary definitions and criteria. Clin Pharm Ther2000; 67: 147.
- 2. Henderson JD, Esham RH. Generic substitution: issues for problematic drugs. South Med J2001; 94: 16–21.
- 3. Murphy JE. Epistaxis associated with elevation of INR in a patient switched to generic warfarin–a comment. Pharmacotherapy2000; 20: 863–5; discussion 869–71. http://doi.org/10.1592/phco.20.9.863.35191
- 4. Murphy JE. Generic substitution and optimal patient care. Arch Intern Med1999; 159: 429–33. http://doi.org/10.1001/archinte.159.5.429
- 5. Meibohm B, Zhang W, Beierle I, et al Epistaxis associated with elevation of INR in a patient switched to generic warfarin-another view. Pharmacotherapy2000; 20: 863–65.
- 6. Swenson CN, Fundak G. Observational cohort study of switching warfarin sodium products in a managed care organization. Am J Health Syst Pharm2000; 57: 452–5. http://doi.org/10.1093/ajhp/57.5.452
- 7. Meyer M, Chan K, Bolton S. Generic warfarin: implications for patient care–another view. Pharmacotherapy1998; 18: 884–6; discussion 887–9.
- 8. Meyer MC. Generic drug product equivalence: current status. Am J Managed Care1998; 4: 1183–91.
- 9. Wagner JL, Dent LA. Epistaxis associated with elevation of INR in a patient switched to generic warfarin. Pharmacotherapy2000; 20: 240–3. http://doi.org/10.1592/phco.20.3.240.34784
- 10. Benson SR, Vance-Bryan K. In favor of Coumadin over generic warfarin. Am J Health Syst Pharm1998; 55: 727–9. http://doi.org/10.1093/ajhp/55.7.727
- 11. Wittkoswsky AK. Generic warfarin: implications for patient care. Pharmacotherapy1997; 17: 640–43.
- 12. Wittkoswsky AK. Generic warfarin: implications for patient care-author’s reply. Pharmacotherapy1998; 18: 887–9.
- 13. Hope KA, Havrda DE. Subtherapeutic INR values associated with a switch to generic warfarin. Ann Pharmacother2001; 35: 183–7. http://doi.org/10.1345/aph.10207
- 14. Check W. Does generic warfarin measure up?CAP Today (College Am Pathologists)1999; 13: 1, 30, 34–7.
- 15. Banahan BF, Bonnarens JK, Bentley JP. Generic substitution of NTI drugs: issues for formulary committee consideration. Formulary1998; 33: 1082–96.
- 16. Haines ST. Reflections on generic warfarin. Am J Health Syst Pharm1998; 55: 729–33. http://doi.org/10.1093/ajhp/55.7.729
- 17. Vercaigne LM, Zhanel GG. Clinical significance of bioequivalence and interchangeability on narrow therapeutic index drugs: focus on warfarins. J Pharm Pharmaceut Sci1998; 1: 92–4.
- 18. Palylyk-Colwell E, Jamali F, Dryden W, et al Bioequivalence and interchangeability of narrow therapeutic range drugs. Canadian Society for Pharmaceutical Sciences discussion. J Pharm Pharm Sci1998; 1: 2–7.
- 19. DeCara JM, Croze S, Falk RH. Generic warfarin: a cost-effective alternative to brand-name drug or a clinical wild card?Chest1998; 113: 261–3. http://doi.org/10.1378/chest.113.2.261
- 20. Pang DC. Drugs with a narrow therapeutic index. American Association of Pharmaceutical Scientists (AAPS) Newsletter1997: 10–13.
- 21. Yacobi A, Masson E, Moros D, et al Who needs individual bioequivalence studies for narrow therapeutic index drugs? A case for warfarin. J Clin Pharmacol2000; 40: 826–35. http://doi.org/10.1177/00912700022009558
- 22. FDA position on product on product selection for narrow therapeutic index drugs (Legal Notes). Am J Health Syst Pharm1997; 56: 615–21.
- 23. Nightengale SL. From the food and drug administration. JAMA1998279: 645.
- 24.
- 25.
- 26. Williams RL. FDA position on product selection for “narrow therapeutic index drugs”. Am J Health Syst Pharm1997; 54: 1630–32.
- 27. Meyer BM. FDA comments on activities in states concerning narrow-therapeutic-index drugs. Am J Health-Syst Pharm1998; 55: 686–7.
- 28. Henney JE. Review of generic bioequivalence studies. JAMA1999; 282: 1995.
- 29.
- 30. Levy G. What are narrow therapeutic index drugs?Clin Pharmacol Ther1998a; 63: 501–5. http://doi.org/10.1016/S0009-9236(98)90100-X
- 31. Hinderling PH. Detection of populations at risk and problem drugs during drug development and in pharmacotherapy. Ther Drug Monit1988; 10: 245–9. http://doi.org/10.1097/00007691-198803000-00001
- 32. Johnston A, Keown PA, Holt DW. Simple bioequivalence criteria: are they relevant to critical dose drugs? Experience gained from cyclosporine. Ther Drug Monit1997; 19: 375–81. http://doi.org/10.1097/00007691-199708000-00002
- 33. Johnston IDA. Difficult drugs to use, few clinical indications. BMJ1990; 300: 929–31.
- 34. Banahan BF 3rd, Kolassa EM. A physician survey on generic drugs and substitution of critical dose medications. Arch Intern Med1997; 157: 2080–8.
- 35.
- 36. Benet LZ. Relevance of pharmacokinetics in narrow therapeutic index drugs. Transplant Proc1999; 31: 1642–4; discussion 1675–84. http://doi.org/10.1016/s0041-1345(99)00083-4
- 37. Benet LZ, Goyan JE. Bioequivalence and narrow therapeutic index drugs. Pharmacotherapy1995; 15: 433–40.
- 38. White RH, Zhou H, Romano P, et al Changes in plasma warfarin levels and variations in steady-state prothrombin times. Clin Pharmacol Ther1995; 58: 588–93. http://doi.org/10.1016/0009-9236(95)90179-5
- 39. Belitsky P, Dunn S, Johnston A, et al Impact of absorption profiling on efficacy and safety of cyclosporin therapy in transplant recipients. Clin Pharmacokinet2000; 39: 117–25. http://doi.org/10.2165/00003088-200039020-00003
- 40. Christians U, First MR, Benet LZ. Recommendations for bioequivalence testing of cyclosporine generics revisited. Ther Drug Monit2000; 22: 330–45. http://doi.org/10.1097/00007691-200006000-00017
- 41. Dumont RJ, Ensom MH. Methods for clinical monitoring of cyclosporin in transplant patients. Clin Pharmacokinet2000; 38: 427–47. http://doi.org/10.2165/00003088-200038050-00004
- 42. Coyle JD, Lima JJ. Procainamide. In: Evans WE, Schentag JJ, Jusko WJ (eds.), Applied Pharmacokinetics, Principles of Therapeutic Drug Monitoring. 3rd ed. Vancouver, WA: Applied Therapeutics, 1992; 22.2–22.33.
- 43. Armijo JA, Cavada E. Graphic estimation of phenytoin dose in adults and children. Ther Drug Monit1991; 13: 507–10. http://doi.org/10.1097/00007691-199111000-00007
- 44. Tozer TN, Winter ME. Phenytoin. In: Evans WE, Schentag JJ, Jusko WJ (eds.), Applied Pharmacokinetics, Principles of Therapeutic Drug Monitoring. 3rd ed. Vancouver, WA: Applied Therapeutics, 1992; 25.1–25.44.
- 45. Patrono C. Aspirin: new cardiovascular uses for an old drug. Am J Med2001; 110: 62–5S. http://doi.org/10.1016/s0002-9343(00)00645-8
- 46. Patrono C, Coller B, Dalen JE, et al Platelet-active drugs: the relationships among dose, effectiveness, and side effects. Chest1998; 114: 470–88S. http://doi.org/10.1378/chest.114.5_supplement.470s
- 47. Hirschowitz BI, Hawkey CJ. Questions regarding future research on aspirin and the gastrointestinal tract. Am J Med2001; 110: 74–8S. http://doi.org/10.1016/s0002-9343(00)00648-3
- 48. Vane JR, Flower RJ, Botting RM. History of aspirin and its mechanism of action. Stoke1990; 21: IV12–IV23.
- 49.
- 50. Yu LX, Jiang W, Zhang X, et al Novel bioequivalence approach for narrow therapeutic index drugs. Clin Pharmacol Ther2015; 97: 286–91. http://doi.org/10.1002/cpt.28
- 51. Baumgartel C, Godman B. Bioequivalence of narrow therapeutic index drugs and immunosuppressives. GaBI J2015: 4: 159–72.
- 52. Blix HS, Viktil KK, Moger TA, et al Drugs with narrow therapeutic index as indicators in the risk of management of hospitalized patients. Pharmacy Practice (Granda)2010; 8: 50–55.
